The CellSearch CTC Enumeration Protocol: A Complete Guide for Cancer Researchers & Drug Developers
This comprehensive guide details the CellSearch system protocol for circulating tumor cell (CTC) enumeration in metastatic cancer, a critical tool for clinical research and therapeutic development.
The CellSearch CTC Enumeration Protocol: A Complete Guide for Cancer Researchers & Drug Developers
Abstract
This comprehensive guide details the CellSearch system protocol for circulating tumor cell (CTC) enumeration in metastatic cancer, a critical tool for clinical research and therapeutic development. It provides foundational knowledge on CTC biology and the CellSearch platform's FDA-cleared methodology. The article offers a step-by-step workflow, from blood sample collection to final CTC identification and enumeration, alongside best practices for consistent results. It addresses common troubleshooting scenarios and optimization strategies for diverse cancer types. Finally, it validates the protocol's clinical utility by comparing its prognostic power and technical performance against emerging CTC technologies, establishing its current role as the gold standard in liquid biopsy for metastatic disease monitoring.
Understanding CTCs and the CellSearch System: The Gold Standard for Metastatic Cancer Enumeration
What are Circulating Tumor Cells (CTCs) and Why Do They Matter in Metastatic Cancer?
Circulating Tumor Cells (CTCs) are malignant cells that have detached from a primary tumor, intravasated into the bloodstream or lymphatic system, and circulate as potential precursors to metastatic deposits. Their presence and enumeration are clinically significant, as they offer a "liquid biopsy" to monitor cancer progression, assess treatment efficacy, and understand the biology of metastasis in real-time. In metastatic cancer research, CTC analysis provides a minimally invasive window into tumor heterogeneity and evolution.
Table 1: Prognostic Value of CTC Enumeration in Metastatic Cancers (CellSearch System)
| Cancer Type | Prognostic Cutoff (CTCs/7.5 mL blood) | Association with Outcome | Key Study/Reference |
|---|---|---|---|
| Metastatic Breast Cancer (MBC) | ≥5 | Shorter Progression-Free Survival (PFS) & Overall Survival (OS) | SWOG S0500 |
| Metastatic Colorectal Cancer (mCRC) | ≥3 | Shorter PFS & OS | CRYSTAL Trial Analysis |
| Metastatic Prostate Cancer (mCRPC) | ≥5 | Shorter OS | IMMC38 Trial |
| Metastatic Non-Small Cell Lung Cancer (NSCLC) | ≥5 | Shorter PFS & OS | SWOG S0425 |
Table 2: Comparison of CTC Analysis Platforms
| Platform/Technology | Detection Principle | Key Advantage | Key Limitation |
|---|---|---|---|
| CellSearch (FDA-cleared) | Immunomagnetic EpCAM-based enrichment, CK/DAPI+/CD45- staining | Standardized, reproducible enumeration; prognostic validation | EpCAM-dependent; no downstream molecular analysis from cartridge. |
| Microfluidic Devices (e.g., CTC-iChip) | Size/deformability or label-free enrichment | EpCAM-independent capture; viable cells for culture | Less standardized; operational complexity. |
| RT-PCR Based Assays | mRNA expression of tumor-associated markers | High sensitivity; multiplex capability | Does not provide intact cells for morphological analysis. |
Application Notes & Protocols: CTC Enumeration via CellSearch System
This protocol is framed within the thesis context of standardizing CTC enumeration for longitudinal monitoring in metastatic cancer research and drug development trials.
Sample Collection and Preparation Protocol
Objective: To collect peripheral blood samples with preserved CTC integrity for CellSearch analysis. Critical Notes: CTCs are fragile and easily lost or degraded; strict adherence to pre-analytical conditions is mandatory.
- Materials: CellSave Preservative Tubes (10mL, containing EDTA and a cellular preservative).
- Procedure:
- Draw blood via venipuncture using a 21-gauge or larger needle.
- Discard the first 2-3 mL of blood to avoid skin cell contamination.
- Collect exactly 7.5 mL of blood directly into the CellSave Preservative Tube.
- Invert the tube gently 8-10 times immediately to mix blood with the preservative. Do not vortex or shake vigorously.
- Store tubes at room temperature (20-25°C). Do not refrigerate or freeze.
- Samples must be processed within 96 hours of blood draw.
CellSearch CTC Enumeration Assay Protocol
Objective: To immunomagnetically isolate, fluorescently label, and enumerate CTCs from 7.5 mL of blood.
Research Reagent Solutions & Essential Materials:
| Item | Function |
|---|---|
| CellSave Preservative Tubes | Stabilizes blood cells and prevents epithelial cell adhesion molecule (EpCAM) degradation. |
| CellSearch CTC Kit | Includes ferrofluid nanoparticles conjugated to anti-EpCAM antibodies for enrichment. |
| CellSearch Staining Reagents | Contains fluorescent monoclonal antibodies: anti-CK-PE (cytokeratin), anti-CD45-APC (leukocyte marker), and the nuclear dye DAPI. |
| CellSearch System (AutoPrep, MagNest, Analyzer II) | Automated platform for sample preparation, immunomagnetic separation, and fluorescence microscopy. |
| CellTracks Analyzer II | Semi-automated fluorescence microscope for CTC identification and enumeration by trained personnel. |
Detailed Workflow:
- Sample Loading: Place the blood tube and the requisite reagent kit onto the CellTrack AutoPrep system.
- Immunomagnetic Enrichment:
- The system centrifuges the sample to separate plasma and cells.
- The cell pellet is resuspended, and anti-EpCAM ferrofluid is added.
- The sample is incubated, allowing magnetic particles to bind to EpCAM-expressing cells (including CTCs).
- A magnetic field is applied to separate labeled cells from unlabeled blood cells.
- Fluorescent Staining:
- Enriched cells are stained with fluorescent antibodies: PE-labeled anti-cytokeratin (CK) to identify epithelial cells, APC-labeled anti-CD45 to identify leukocytes, and DAPI to stain nuclei.
- Cells are permeabilized to allow intracellular CK staining.
- Magnetic Cartridge Assembly:
- The stained sample is pumped into a MagNest cell presentation cartridge.
- The cartridge is placed in a magnetic field, drawing magnetically labeled cells to the surface of a glass slide within the cartridge.
- Enumeration & Analysis:
- The cartridge is transferred to the CellTracks Analyzer II, a 4-color semi-automated fluorescence microscope.
- The instrument scans the cartridge surface and captures images of all fluorescent events.
- A trained analyst reviews images according to standardized criteria:
- A CTC is defined as a DAPI+ nucleated cell, CK-positive (fluorescent), and CD45-negative.
- White blood cells are DAPI+, CK-negative, and CD45+.
- Debris or artifacts are typically DAPI negative.
Downstream Molecular Characterization Protocol (Post-Enumeration)
Objective: To perform genomic or protein expression analysis on CTCs enumerated via CellSearch.
Note: This is an advanced, research-grade protocol. The standard CellSearch cartridge is not designed for this; specialized retrieval methods are required.
- CTC Retrieval from Magnest Cartridge:
- Carefully disassemble the MagNest cartridge after scanning.
- Using a phosphate-buffered saline (PBS) wash and a fine pipette, gently flush the surface of the glass slide to collect cells into a microcentrifuge tube.
- Cell Lysis and Nucleic Acid Isolation:
- Centrifuge the cell suspension. Lyse the pellet using a commercial lysis buffer containing Proteinase K.
- Extract DNA or RNA using silica-column or magnetic bead-based kits optimized for low-input samples (e.g., QIAamp DNA Micro Kit).
- Downstream Analysis:
- For DNA: Perform whole-genome amplification (WGA) followed by next-generation sequencing (NGS) for mutations (e.g., ESR1, PIK3CA) or digital PCR for specific targets.
- For RNA: Perform reverse transcription and pre-amplification, followed by qRT-PCR for gene expression profiling (e.g., AR-V7 in prostate cancer).
Visualizations
CTC Enumeration via CellSearch Workflow
CTC Identification Logic Tree
History and FDA Clearance
The CellSearch system, developed by Veridex LLC (now a part of Menarini Silicon Biosystems), represents the first diagnostic platform for the quantitative enumeration of circulating tumor cells (CTCs) in whole blood. Its clinical validation and subsequent FDA clearances established a new paradigm in metastatic cancer monitoring.
Table 1: History and Key FDA Clearances of the CellSearch System
| Year | Milestone | Significance |
|---|---|---|
| 2004 | Initial FDA Clearance (510(k)) | Cleared for monitoring CTCs in metastatic breast cancer (MBC). First-ever FDA-cleared CTC test. |
| 2007 | FDA Clearance Expanded | Cleared for monitoring CTCs in metastatic colorectal cancer (mCRC). |
| 2008 | FDA Clearance Expanded | Cleared for monitoring CTCs in metastatic prostate cancer (mCRPC). |
| 2016-2022 | Continued Market Availability & Updates | System remains the gold standard; software and kit component improvements are implemented. |
Role as a Clinical Research Tool
Beyond prognostication, CellSearch has become a cornerstone tool in translational oncology research. It enables non-invasive, serial "liquid biopsies" to study cancer biology, track clonal evolution, and assess pharmacodynamic responses to therapy in real time. Its primary role in research includes:
- Patient Stratification: Using baseline CTC counts as a prognostic biomarker for clinical trial enrollment.
- Early Efficacy Assessment: Monitoring CTC changes as a surrogate endpoint for therapy response, potentially preceding radiographic changes.
- Drug Development: Serving as a pharmacodynamic biomarker to confirm target engagement of novel therapeutics.
- Molecular Characterization: Isolated CTCs can be used for downstream molecular analyses (e.g., FISH, protein expression, rare cell sequencing).
Application Notes & Protocols for CTC Enumeration in Metastatic Cancer Research
Core Principle
The CellSearch system immunomagnetically enriches and fluorescently identifies epithelial-derived CTCs from whole blood. It targets cells expressing the Epithelial Cell Adhesion Molecule (EpCAM) for capture, and uses fluorescent stains for cytokeratins (CK 8, 18, 19), CD45, and DAPI to define a CTC: CK+, DAPI+, CD45-.
Detailed Protocol for CTC Enumeration
Aim: To enumerate CTCs from 7.5 mL of peripheral blood drawn from patients with metastatic breast, colorectal, or prostate cancer.
I. Materials & Pre-Analytical Steps
Research Reagent Solutions & Essential Materials
| Item | Function |
|---|---|
| CellSave Preservative Tubes | Contains EDTA and a cellular preservative. Stabilizes blood samples for up to 96 hours post-draw to prevent degradation of CTCs. |
| CellSearch CTC Kit | Disease-specific kit (e.g., Breast, CRC, Prostate). Contains ferrofluid nanoparticles conjugated to anti-EpCAM antibodies, staining reagents (anti-CK-PE, anti-CD45-APC), permeabilization buffer, and nuclear dye (DAPI). |
| CellSearch AutoPrep System | Automated sample processing platform. Performs immunomagnetic enrichment, staining, washing, and cartridge preparation. |
| CellSearch Magnest | Magnetic assembly that holds the cartridge during processing to retain magnetically labeled cells. |
| CellSearch Cartridge | Microfluidic chamber where enriched cells are mounted for analysis. |
| CellTracks Analyzer II | Semi-automated fluorescence microscope. Digitally scans the cartridge and presents image galleries for final classification by a trained operator. |
| Control Materials | Kit controls (positive and negative) for system performance verification. |
Sample Collection:
- Draw blood into CellSave Preservative Tubes.
- Invert tube gently 8 times to mix.
- Store at room temperature (15-30°C).
- Process within 96 hours of blood draw.
II. Automated Sample Preparation (CellSearch AutoPrep)
- Tube Placement: Load patient samples, controls, and buffers into designated positions on the AutoPrep deck.
- Run Initiation: Select the appropriate assay protocol. The system performs:
- Immunomagnetic Enrichment: Incubates sample with anti-EpCAM ferrofluid. Magnetically captures labeled cells.
- Washing: Removes unbound cells and plasma components.
- Staining: Permeabilizes cells and incubates with fluorescent monoclonal antibodies (anti-CK-PE, anti-CD45-APC) and DAPI.
- Resuspension & Transfer: Magnetically concentrated cells are resuspended and pumped into a CellSearch Cartridge, which is placed in the Magnest.
III. Analysis and Interpretation (CellTracks Analyzer II)
- The cartridge is scanned by the Analyzer, which uses a 4-laser system to detect DAPI, PE (CK), and APC (CD45) fluorescence.
- Software compiles images of all objects meeting pre-set fluorescence thresholds.
- Operator Classification: A trained scientist reviews image galleries to classify objects according to predefined criteria:
- CTC: DAPI+, CK-PE+, CD45-APC-, morphologically intact.
- Leukocyte: DAPI+, CK-PE-, CD45-APC+.
- Debris/Other: All other events (e.g., CK-, CD45-; irregular DAPI).
- The final CTC count per 7.5 mL of blood is reported.
Table 2: Key Quantitative Parameters and Clinical Cut-Offs
| Parameter | Specification | Clinical/Research Significance |
|---|---|---|
| Sample Volume | 7.5 mL | Standardized volume for all assays. |
| Detection Limit | 1-2 CTCs/7.5 mL | High sensitivity for rare cell detection. |
| Established Prognostic Cut-off | ≥5 CTCs/7.5 mL (MBC, mCRPC) | Baseline count predicting shorter PFS and OS. ≥3 CTCs/7.5 mL for mCRC. |
| Analytical Time | ~6.5 hours (hands-on ~0.5h) | Enables batch processing. |
| Assay Precision | High (CV <20% for samples with >10 CTCs) | Reliable for serial monitoring. |
Protocol for Downstream Molecular Analysis (Research-Use Only)
Aim: To perform fluorescence in situ hybridization (FISH) on CTCs enumerated via CellSearch for research purposes.
Method:
- After scanning and before disassembling the cartridge, map the X-Y coordinates of identified CTCs.
- Carefully disassemble the cartridge and transfer the membrane containing the cells to a glass slide.
- Follow standard FISH protocol:
- Apply target-specific FISH probes (e.g., for HER2 amplification in breast cancer).
- Denature and hybridize.
- Wash stringently.
- Mount with DAPI-containing medium.
- Relocate the pre-identified CTCs using the coordinate map on a standard fluorescence microscope.
- Score FISH signals specifically within the morphologically defined CTCs.
Visualizations
Title: CellSearch CTC Enumeration Workflow
Title: Roles of CTCs in Cancer Research
Within the broader thesis on the standardized CellSearch system protocol for Circulating Tumor Cell (CTC) enumeration in metastatic cancer research, this document details the core technological components. The CellSearch system is an FDA-cleared, semi-automated platform that reproducibly identifies and enumerates CTCs from peripheral blood. Its clinical utility in prognosticating progression-free and overall survival in metastatic breast, colorectal, and prostate cancers is well-established. The technology's foundation is the sequential application of EpCAM-based immunomagnetic enrichment followed by multi-channel fluorescent staining, which together enable the specific isolation and positive identification of rare CTCs against a background of billions of hematopoietic cells.
The process leverages the frequent overexpression of the Epithelial Cell Adhesion Molecule (EpCAM) on carcinomas. Blood samples are first enriched for EpCAM-expressing cells using ferrofluid nanoparticles conjugated with anti-EpCAM antibodies. This immunomagnetic capture concentrates potential CTCs. Subsequently, the enriched sample is stained with fluorescent reagents for nuclei (DAPI), leukocytes (CD45-APC), and epithelial cytokeratins (CK-PE). The definitive identification of a CTC is based on a consensus phenotype: DAPI+/CK+/CD45-.
Key Research Reagent Solutions
The following table lists essential materials and reagents central to the CellSearch protocol and related research applications.
| Item Name | Function & Explanation |
|---|---|
| CellSave Preservative Tubes | Vacutainer tubes containing EDTA and a cellular preservative. Prevents coagulation and stabilizes epithelial cell surface markers for up to 96 hours post-blood draw, critical for sample integrity. |
| CellSearch Epithelial Cell Kit | Kit includes: Anti-EpCAM ferrofluid, Staining reagents (CK-PE, CD45-APC, DAPI), Permeabilization buffer, and Sample containers. Provides all standardized reagents for the CTC capture and identification workflow. |
| CellSearch System (AutoPrep & Analyzer) | The AutoPrep automates sample preparation, immunomagnetic enrichment, and staining. The Analyzer is a semi-automated fluorescence microscope for scanning and presenting digital images for review. |
| Anti-EpCAM Ferrofluid | Colloidal magnetic nanoparticles (~100-200 nm) conjugated to monoclonal antibodies against EpCAM. Enables magnetic separation of EpCAM+ cells from the bulk blood sample. |
| CK-PE (Cytokeratin 8, 18, 19) | A pan-cytokeratin antibody cocktail conjugated to Phycoerythrin (PE). Fluorescently labels the intracellular cytokeratin network of epithelial-derived CTCs (emission ~575 nm). |
| CD45-APC | Antibody against the common leukocyte antigen CD45 conjugated to Allophycocyanin (APC). Labels all white blood cells, serving as a negative selection marker to exclude leukocytes from CTC counts (emission ~660 nm). |
| 4',6-Diamidino-2-Phenylindole (DAPI) | A cell-permeant, DNA-binding fluorescent dye. Stains the nuclei of all nucleated cells, allowing for cell localization and assessment of nuclear morphology (emission ~460 nm). |
| Magnetic Particle Concentrator (MPC) | A rack that creates a high-gradient magnetic field. Used during manual protocol steps to hold ferrofluid-bound cells against the tube wall while undesired components are aspirated. |
Detailed Protocol: CellSearch CTC Enumeration
Principle: Isolate and enumerate CTCs (DAPI+/CK+/CD45-) from 7.5 mL of peripheral blood. Sample: Blood drawn into CellSave tubes. Process within 96 hours.
Step-by-Step Methodology
Sample Preparation:
- Centrifuge the 7.5 mL blood sample at 800 x g for 10 minutes at room temperature.
- Transfer plasma to a new tube, leaving ~1.0 mL of plasma with the cell pellet.
- Resuspend the cell pellet completely.
Immunomagnetic Enrichment:
- Add 6.5 µL of anti-EpCAM ferrofluid to the sample.
- Incubate for 10 minutes at room temperature with gentle tilting and rotation to allow antibody binding.
- Place the sample tube in the AutoPrep system or a manual MPC for 10 minutes to capture labeled cells.
- Aspirate and discard the supernatant (containing unbound cells and plasma).
- Remove from the magnet and resuspend the captured cells in 1 mL of wash buffer.
Fluorescent Staining & Permeabilization:
- Add the staining reagent mixture (containing CK-PE, CD45-APC, and permeabilization buffer) to the sample.
- Incubate for 20 minutes at room temperature, protected from light.
- Add 2 µg/mL DAPI (final concentration) and incubate for 5-10 minutes.
- Perform a final magnetic capture and resuspend the stained, enriched cell fraction in ~100 µL of assay buffer. Transfer to a MagNest cell presentation cartridge.
Analysis & Enumeration:
- Place the cartridge in the CellTracks Analyzer II.
- The system automatically scans the entire sample surface, capturing fluorescent images in four channels (DAPI, FITC, PE, APC).
- Images are compiled and presented as computer-generated galleries based on initial algorithms (CK-PE signal, nuclear size).
- A trained technician reviews each gallery event to definitively classify CTCs based on the following criteria:
- CTC Positive: Morphologically intact cell, DAPI+, CK-PE+, CD45-APC-.
- Leukocyte: DAPI+, CK-PE-, CD45-APC+.
- Debris/Artifact: Does not meet all CTC criteria.
Key Quantitative Performance Data
Table 1: Typical Performance Characteristics of the CellSearch System.
| Parameter | Value/Range | Notes |
|---|---|---|
| Sample Input Volume | 7.5 mL | Standard for clinical studies. |
| Enrichment Capture Efficiency | >85% (for EpCAM+ cell lines spiked into blood) | Varies with EpCAM expression level and cell type. |
| Average Purity of Enrichment | 0.1% - 1.0% | Ratio of CTCs to total nucleated cells in the final product. Highlights the "needle-in-a-haystack" challenge. |
| Analytical Sensitivity (Limit of Detection) | 1-2 CTCs per 7.5 mL | Defined by low spike-in recovery studies. |
| Inter-operator Reproducibility (CV) | <15% | Due to automated preparation and standardized analysis criteria. |
| Clinical Cut-off (Prognostic) | ≥5 CTCs / 7.5 mL (MBC) | Metastatic Breast Cancer; ≥3 for mCRPC, ≥3 for mCRC. |
Table 2: Fluorescent Channel Specifications for Identification.
| Marker | Fluorochrome | Excitation (nm) | Emission (nm) | Purpose |
|---|---|---|---|---|
| Nuclear Stain | DAPI | 358 | 461 | Identifies nucleated cells. |
| Cytokeratin (CK) | Phycoerythrin (PE) | 496 | 578 | Positive marker for epithelial origin (CTCs). |
| Leukocyte (CD45) | Allophycocyanin (APC) | 650 | 660 | Negative marker; excludes WBCs. |
| System Autofluorescence | FITC Channel | 496 | 516 | Used for system alignment and background check. |
Diagrams
Core CTC Identification Logic
EpCAM Signaling & Capture Rationale
Within the framework of a thesis on the CellSearch system protocol for CTC enumeration in metastatic cancer research, the precise immunophenotypic definition of a Circulating Tumor Cell (CTC) is paramount. The CellSearch system (Menarini Silicon Biosystems) is the first and only FDA-cleared method for the automated detection and enumeration of CTCs as a prognostic biomarker. In metastatic carcinomas of epithelial origin (e.g., breast, prostate, colorectal), CTCs are defined by a specific triplet of markers: positive for cytokeratins (CK) and a nuclear stain (DAPI), and negative for the common leukocyte antigen CD45.
Core Definition & Rationale
The canonical CellSearch CTC is defined by the phenotype: CK+, DAPI+, CD45-. This definition is based on robust immunofluorescent staining and digital imaging.
| Marker | Fluorochrome | Expression Purpose | Interpretation in CTC Definition |
|---|---|---|---|
| Cytokeratins (CK 8, 18, 19) | Phycoerythrin (PE) | Positive Selection | Identifies cells of epithelial origin. CKs are intermediate filaments expressed in epithelial cells and carcinomas. |
| CD45 | Allophycocyanin (APC) | Negative Selection | Identifies leukocytes. Its absence excludes contaminating white blood cells from the CTC count. |
| DAPI | DAPI | Positive Selection | A nuclear stain confirming the presence of an intact nucleus, excluding cellular debris and anucleate events. |
Additional Morphological Criteria: Beyond fluorescence, qualified CTCs must exhibit morphological characteristics consistent with a cell, including a round-to-oval morphology, a nucleus occupying >75% of the cytoplasmic area, and a CK staining pattern that is circumferential and granular.
Detailed Experimental Protocol for CTC Enumeration via CellSearch
Principle: Immunomagnetic enrichment of epithelial cells from whole blood followed by immunofluorescent staining and semi-automated digital microscopy.
Materials & Reagent Solutions
Research Reagent Solutions Toolkit:
| Reagent / Material | Function in Protocol |
|---|---|
| CellSave Preservative Tubes | Blood collection tubes containing EDTA and a cellular preservative. Stabilizes blood samples for up to 96 hours pre-processing. |
| CELLection Dynabeads | Magnetic beads coated with EpCAM (Epithelial Cell Adhesion Molecule) antibody. Enriches epithelial cells from the blood sample. |
| Staining Reagent: Anti-CK-PE | Monoclonal antibody cocktail (C11, A53-B/A2) targeting CK 8, 18, 19, conjugated to PE. Labels epithelial cytoplasm. |
| Staining Reagent: Anti-CD45-APC | Monoclonal antibody (HI30) targeting CD45, conjugated to APC. Labels leukocytes. |
| Staining Reagent: DAPI | Nucleic acid dye. Labels the nucleus. |
| Permeabilization Buffer | Permeabilizes cells to allow CK antibodies to enter. |
| CellSearch Assay Kit | Proprietary kit containing all necessary buffers, staining reagents, and capture tubes. |
| CellTracks AutoPrep System | Automated sample preparation system for immunomagnetic enrichment, staining, and aspiration. |
| CellTracks Analyzer II | Semi-automated 4-color fluorescence microscope. Digitally scans the sample cartridge and presents galleries of potential CTCs for final classification. |
Step-by-Step Protocol
- Sample Collection: Collect 7.5 mL of venous blood directly into a CellSave tube. Invert gently 8 times. Store at room temperature (15-30°C). Process within 96 hours of draw.
- Sample Setup: Pipette 7.5 mL of blood into the CellSearch AutoPrep sample tube. Load the tube, along with the necessary assay kit and reagent strips, onto the CellTracks AutoPrep system.
- Automated Processing (AutoPrep): a. Immunomagnetic Enrichment: The system adds anti-EpCAM ferrofluid to the sample. EpCAM-positive cells (epithelial cells) are captured onto magnetic beads. b. Washing & Transfer: The magnetically labeled cells are washed and transferred to a microscope cartridge (the "MagNest"). c. Staining: The system sequentially adds Permeabilization Buffer, anti-CK-PE, anti-CD45-APC, and DAPI to stain the immobilized cells. d. Sealing: The cartridge is sealed and is now ready for analysis.
- Image Acquisition & Analysis (Analyzer II): a. The MagNest cartridge is placed into the CellTracks Analyzer II. b. The instrument uses magnets to position the immunomagnetically captured cells into a single focal plane. c. It automatically scans the entire sample surface using a 10x objective, capturing digital images in four fluorescent channels (DAPI, FITC, PE, APC).
- CTC Classification & Enumeration: a. Proprietary software algorithms identify objects that are DAPI-positive and CK-PE-positive. b. These objects are presented in a browser-based gallery for operator review. c. The operator manually confirms each potential CTC based on the strict criteria: CK+ (red), DAPI+ (blue), CD45- (no green signal), and appropriate morphology. d. Events that are DAPI+, CK-, CD45+ are classified as leukocytes. Events that are DAPI+, CK+, CD45+ are classified as dual-staining events (rare, typically ignored). e. The final CTC count is reported as the number of confirmed CTCs per 7.5 mL of blood.
Data Interpretation & Quantitative Thresholds
The prognostic utility of CTC enumeration is tied to validated clinical thresholds.
| Cancer Type (Metastatic) | Clinically Significant Cut-off | Prognostic Meaning |
|---|---|---|
| Breast | ≥5 CTCs / 7.5 mL | Shorter Progression-Free Survival (PFS) and Overall Survival (OS). |
| Prostate | ≥5 CTCs / 7.5 mL | Shorter OS. Used for risk stratification. |
| Colorectal | ≥3 CTCs / 7.5 mL | Shorter PFS and OS. |
Important Notes: A count below the threshold is considered "favorable." The continuous variable (actual CTC count) also holds prognostic and potential pharmacodynamic information in clinical trials.
Visualization of CTC Identification Logic & Workflow
Title: CTC Identification Logic Flow on CellSearch
Title: CellSearch CTC Enumeration Protocol Workflow
The enumeration of Circulating Tumor Cells (CTCs) via the CellSearch system provides a critical, minimally invasive biomarker for assessing prognosis and treatment efficacy in metastatic cancer. Within the context of a broader thesis on CTC enumeration protocols, this document outlines the approved clinical indications and key investigational applications of CTC analysis in metastatic breast (MBC), prostate (mCRPC), and colorectal cancer (mCRC), detailing the experimental methodologies that underpin these uses.
Approved Clinical Indications & Key Quantitative Findings
The CellSearch system is cleared by the FDA for specific prognostic indications in metastatic breast, prostate, and colorectal cancers. The table below summarizes the validated prognostic thresholds and their clinical implications.
Table 1: FDA-Cleared Prognostic Indications for CellSearch CTC Enumeration
| Cancer Type | Approved Indication | Key Prognostic Cut-off (CTCs/7.5mL) | Clinical Implication |
|---|---|---|---|
| Metastatic Breast Cancer (MBC) | Prognostic marker pre-therapy and during therapy. | ≥5 | Shorter Progression-Free Survival (PFS) and Overall Survival (OS). |
| Metastatic Castration-Resistant Prostate Cancer (mCRPC) | Prognostic marker pre-therapy and during therapy. | ≥5 | Shorter OS. Independent predictor of survival. |
| Metastatic Colorectal Cancer (mCRC) | Prognostic marker pre-therapy. | ≥3 | Shorter PFS and OS. |
Investigational Applications & Protocol Designs
Beyond prognosis, CTC enumeration and characterization are pivotal in investigational studies for monitoring treatment response and drug development.
Application Note 1: Early Treatment Response Assessment in mCRPC
- Objective: To determine if a decrease in CTC count after initiating a novel androgen receptor pathway inhibitor (ARPI) correlates with radiographic PFS.
- Protocol:
- Baseline Sample: Collect 2x 10mL blood samples into CellSave Preservative Tubes from mCRPC patients prior to Cycle 1, Day 1 of therapy.
- On-Treatment Sample: Repeat blood collection at Cycle 3, Day 1.
- CTC Enumeration: Process samples per the validated CellSearch CTC Kit (for enumeration). Briefly:
- Immunomagnetic enrichment using ferrofluid nanoparticles coated with anti-EpCAM antibodies.
- Staining with fluorescently labeled anti-cytokeratin (CK 8, 18, 19) antibodies (CTC identifier), anti-CD45 (leukocyte exclusion), and DAPI (nuclear stain).
- Automated fluorescence microscopy and manual review to identify CTCs (CK+/DAPI+/CD45-).
- Data Analysis: Categorize patients as "CTC Responders" (conversion from ≥5 to <5 CTCs) or "Non-Responders." Compare radiographic PFS between groups using Kaplan-Meier analysis and log-rank test.
Application Note 2: Phenotypic Subtyping in MBC for Therapy Guidance
- Objective: To investigate the prevalence of HER2+ CTCs in patients with HER2-negative primary breast cancer to assess potential for targeted therapy.
- Protocol:
- Sample Collection: Draw blood from metastatic HER2-negative MBC patients into CellSave tubes.
- CTC Enumeration & HER2 Staining: Use the CellSearch CTC Kit with a modified staining panel. After standard CK/CD45/DAPI staining, add a fluorescent anti-HER2 antibody.
- Image Acquisition & Analysis: Use the CellTracks Analyzer II to capture images. During review, classify CTCs as HER2-positive or -negative based on membrane fluorescence intensity above a pre-defined threshold.
- Correlative Analysis: Correlate the presence of HER2+ CTCs with patient demographics, site of metastasis, and response to subsequent therapies in observational studies.
Application Note 3: Assessing EMT in mCRC for Prognostic Stratification
- Objective: To evaluate the presence of CTCs with a mesenchymal phenotype (partial EMT) and their association with resistance to anti-EGFR therapy in RAS wild-type mCRC.
- Protocol:
- Patient Cohort: RAS wild-type mCRC patients initiating cetuximab + chemotherapy.
- Sample Processing: Enrich CTCs using the CellSearch Profile Kit, which isolates nucleated cells without EpCAM selection, preserving EpCAM-low CTCs.
- Downstream Analysis: Extract mRNA from the enriched cell fraction.
- Gene Expression Profiling: Perform RT-qPCR for epithelial (e.g., EPCAM, CK19) and mesenchymal (e.g., VIM, ZEB1) transcripts.
- Scoring: Calculate an "EMT Score" based on the ratio of mesenchymal to epithelial genes. Correlate high EMT scores with clinical outcomes (PFS/OS).
Visualization of Key Concepts
Title: CellSearch CTC Enumeration Workflow
Title: AR Pathway & Drug Target in mCRPC
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for CellSearch-Based Investigational Studies
| Item | Function |
|---|---|
| CellSave Preservative Blood Collection Tube | Maintains CTC integrity and prevents epithelial cell adhesion molecule (EpCAM) degradation for up to 96 hours post-draw. |
| CellSearch CTC Kit (for enumeration) | Provides all reagents (ferrofluid, staining antibodies, buffers) for the standardized enumeration of epithelial CTCs. |
| CellSearch Profile Kit | Enriches all nucleated cells without epithelial selection, enabling downstream molecular analysis of EpCAM-low CTCs. |
| CellSearch CXC Kit | For capturing circulating endothelial cells (CECs) and other rare cell types in investigational oncology studies. |
| Anti-HER2 (or other protein) Antibody, Fluorescent Conjugate | For phenotypic characterization of CTCs beyond the standard epithelial/leukocyte markers. |
| CellTracks Magnest | The semi-automated instrument system for immunomagnetic enrichment and sample preparation. |
| CellTracks Analyzer II | A four-color fluorescence microscope for automated digital imaging of prepared samples. |
| RNA Stabilization Reagent (e.g., RNAlater) | For stabilizing nucleic acids in CTCs enriched via the Profile Kit for subsequent PCR or sequencing. |
Step-by-Step CellSearch Protocol: From Blood Draw to CTC Enumeration
Within the context of a broader thesis on the CellSearch system for Circulating Tumor Cell (CTC) enumeration in metastatic cancer research, the pre-analytical phase is the most critical determinant of data integrity and clinical validity. CTCs are rare and fragile, making standardized protocols for blood collection, tube handling, and storage paramount to prevent cell loss, degradation, or artifact generation, which can directly impact patient stratification and therapeutic response assessment in drug development.
The following tables consolidate key quantitative parameters from current literature and manufacturer guidelines essential for CellSearch-based studies.
Table 1: Blood Collection Protocol Specifications
| Parameter | Specification | Rationale |
|---|---|---|
| Draw Volume | 10 mL per CellSave tube. Must be filled to stated volume. | Ensures correct blood-to-preservative ratio for cell stabilization. |
| Tube Type | CellSave Preservative Tubes (or other FDA-cleared/validated tubes). | Contains EDTA and a cellular preservative; prevents clotting and stabilizes epitopes. |
| Tube Mixing | Gently invert tube 8-10 times immediately after draw. | Ensures complete mixing with preservative; prevents clotting. |
| Draw Order | After serum tubes, before tubes with anticoagulants like heparin or EDTA for other tests. | Prevents cross-contamination of anticoagulants that can interfere with CellSearch assay. |
| Time to Processing | ≤96 hours (4 days) from draw at room temp (20-26°C). | Validated stability window for CTC integrity and EpCAM expression. |
Table 2: Impact of Pre-Analytical Deviations on CTC Enumeration
| Deviation | Experimental Impact on CTC Count/Recovery | Reference Range of Effect |
|---|---|---|
| Incomplete Fill | Reduced preservative efficacy; clotting; cell loss. | Can reduce recovery by >50%. |
| Delayed Mixing | Microclot formation; CTCs trapped in clot. | Unquantifiable but significant loss; invalidates sample. |
| Incorrect Storage Temp | Refrigeration (<4°C): cell degradation. Excessive heat: increased apoptosis. | Refrigeration can reduce counts by 30-70% vs. RT. |
| Exposure to Vibration | Potential mechanical stress on cells during transport. | Qualitative increase in non-specific background. |
| Time Beyond 96h | Increased leukocyte degradation, reduced CTC integrity. | Counts may decrease progressively post-96h. |
Detailed Experimental Protocols
Protocol 1: Standardized Venipuncture for CTC Enumeration
- Patient Preparation: Standard patient identification. No special fasting required, but note recent therapies.
- Tourniquet Application: Apply for ≤1 minute. Prolonged stasis may affect cell counts.
- Venipuncture: Perform using a 21-gauge or larger needle. Discard the first 2-3 mL of blood if drawn without a diversion apparatus to avoid skin cell contamination.
- Tube Filling: Draw blood directly into the CellSave Preservative Tube. Allow vacuum to fill tube completely to 10 mL mark.
- Immediate Mixing: Within 3 seconds of draw, gently invert the tube 8-10 times (do not shake vigorously).
- Labeling: Label tube with patient ID, date, and time of collection.
- Storage Post-Collection: Store upright at room temperature (20-26°C) until processing. Do not refrigerate or freeze.
Protocol 2: Validation of Sample Stability for Extended Storage Studies Objective: To empirically verify CTC stability in preservative tubes up to the 96-hour window under simulated transport conditions.
- Sample Collection: Collect blood from metastatic cancer patients (n≥10) into 4 CellSave tubes per patient.
- Time-Point Allocation: Process each tube from the same draw at different time points: T=0h (baseline, ideally within 24h), T=48h, T=72h, T=96h. All tubes stored at 22°C ± 2°C.
- CellSearch Processing: Process each sample according to the FDA-cleared CellSearch protocol: immunomagnetic enrichment with anti-EpCAM ferrofluid, staining with anti-CK-PE (cytokeratin), anti-CD45-APC (leukocyte marker), and DAPI (nuclear stain).
- Analysis: Enumerate CTCs (CK+, DAPI+, CD45-) using the CellTracks Analyzer II. Record total CTCs and morphology metrics.
- Statistical Comparison: Use a non-parametric test (e.g., Wilcoxon signed-rank) to compare CTC counts at each time point (48h, 72h, 96h) against the T=0h baseline. Accept stability if there is no statistically significant decrease (p>0.05) and recovery is within 20% of baseline.
Signaling Pathways and Workflows
CellSearch Pre-Analytical & Analytical Workflow
Impact of Handling on CTC Capture
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in CellSearch Pre-Analytical Phase |
|---|---|
| CellSave Preservative Tube | Proprietary blood collection tube containing EDTA and a fixative. Stabilizes cells, prevents clotting, and preserves surface epitopes (especially EpCAM) for up to 96 hours. |
| CellSearch Circulating Tumor Cell Kit | Includes anti-EpCAM ferrofluid, staining reagents (anti-CK-PE, anti-CD45-APC, DAPI), and assay buffer for the immunomagnetic enrichment and fluorescent staining process. |
| CellTracks AutoPrep System | Automated sample preparation instrument that standardizes the enrichment, staining, and transfer of cells to the cartridge, minimizing manual variability. |
| CellTracks Magnest | Magnetic assembly used during processing to hold magnetically-labeled cells (CTCs) during wash steps. |
| CellSearch Control Kit | Contains stable cell line controls (high/low) for validating instrument performance and assay accuracy in each run. |
| Temperature Monitor | Data logger to ensure continuous room temperature (20-26°C) storage/transport of tubes prior to processing. |
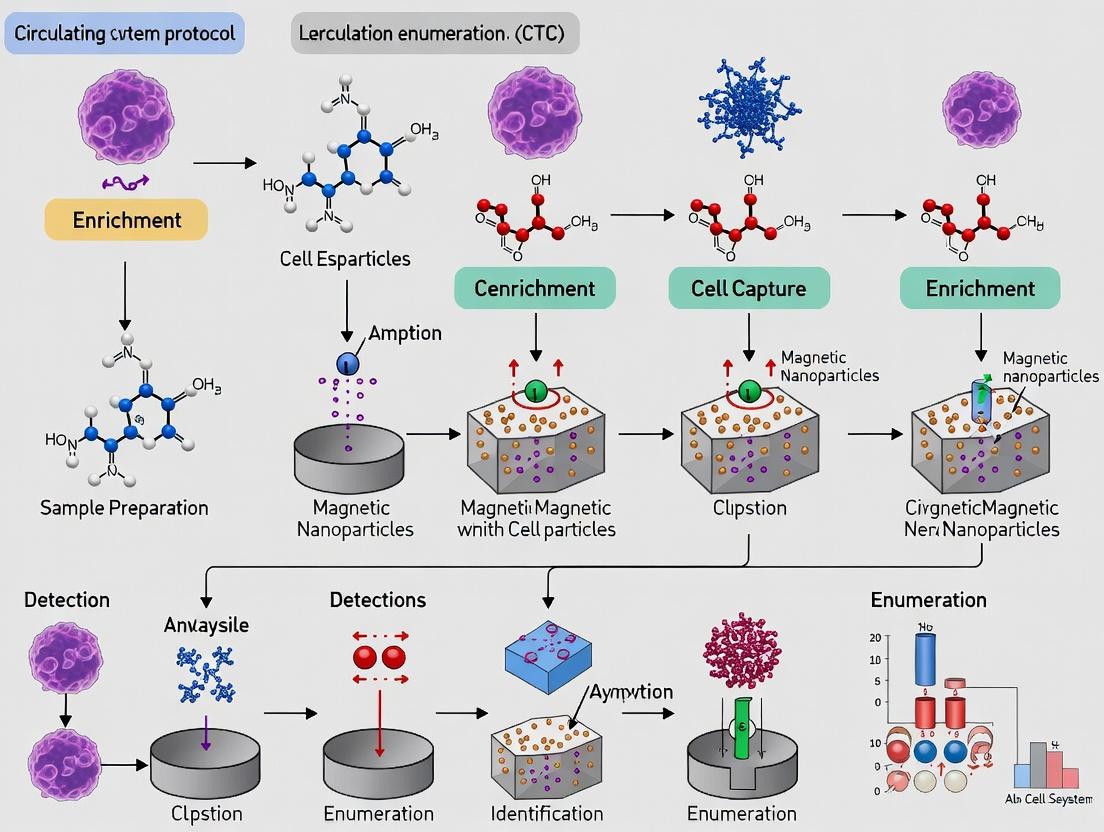
Within the broader thesis on the CellSearch system protocol for Circulating Tumor Cell (CTC) enumeration in metastatic cancer research, the sample preparation and loading phase is a critical pre-analytical determinant of data fidelity. This standardized, FDA-cleared methodology enables the reproducible quantification of epithelial-derived CTCs from whole blood, a validated prognostic biomarker in metastatic breast, colorectal, and prostate cancers. The process hinges on the immunomagnetic selection of cells expressing the Epithelial Cell Adhesion Molecule (EpCAM), followed by fluorescent staining for nuclei (DAPI), cytokeratins 8, 18+, and 19 (CK-PE), and leukocytes (CD45-APC). Accurate loading of the prepared sample into the proprietary cartridge is the final, manual step before automated fluorescence-based analysis on the CellTracks Analyzer II. Consistency here is paramount for minimizing technical variability in longitudinal studies assessing therapeutic response.
Detailed Loading Protocol
Principle: The immunomagnetically enriched and stained cell sample is resuspended in a precise volume of dilution buffer and transferred to the dual-chambered MagNest cell presentation cartridge. This cartridge is engineered to facilitate the magnetic sedimentation of labeled cells into a single focal plane for automated microscopy.
Materials & Equipment:
- Processed sample tube containing magnetically labeled and stained cells.
- CellSearch Dilution Buffer (Part # 7900005).
- CellSearch MagNest Cell Presentation Cartridge (Part # 6400002).
- CellSearch Sample Tube (12 x 75 mm, Part # 9501001).
- Fixed-volume pipettes (100 µL and 150 µL) and sterile tips.
- Vortex mixer.
- Magnetic cartridge rack (from CellSearch Kit).
Methodology:
- Final Resuspension: Following the staining and washing steps, ensure the sample tube is positioned off the magnet. Using a pipette, gently remove all but approximately 100 µL of supernatant. Vortex the sample tube for 3 seconds at medium speed to resuspend the cell pellet.
- Dilution: Add exactly 150 µL of CellSearch Dilution Buffer to the sample tube. Cap the tube and vortex for 3 seconds to achieve a homogeneous cell suspension in a total volume of approximately 250 µL.
- Cartridge Loading: Place a MagNest cartridge in the provided rack.
- Pipette 100 µL of the well-mixed sample suspension into the left chamber (S) of the cartridge.
- Piper another 100 µL into the right chamber (S) of the same cartridge. Avoid introducing air bubbles.
- Preparation for Analysis: Gently place a cover on the loaded cartridge. The cartridge is now ready for insertion into the CellTracks Analyzer II for automated scanning and enumeration.
Table 1: Critical Volumes and Parameters for Sample Loading
| Parameter | Specification | Purpose & Rationale |
|---|---|---|
| Final Supernatant Volume | ~100 µL | Standardizes starting volume before dilution buffer addition to ensure consistent cell concentration. |
| Dilution Buffer Volume | 150 µL | Optimizes cell density and suspension viscosity for even distribution in the cartridge. |
| Total Load Volume per Chamber | 100 µL | Defined by cartridge geometry to create a monolayer in the magnetic imaging region. |
| Total Sample Consumed | 200 µL (of ~250 µL) | Leaves a small residual volume; the process is designed for the dual-chamber load. |
| Time-to-Analysis | ≤ 4 hours post-loading | Prevents degradation of fluorescence signals and maintains cell integrity. |
Table 2: Impact of Loading Errors on Analytical Outcomes
| Loading Deviation | Potential Effect on CTC Enumeration | Corrective Action |
|---|---|---|
| Air bubbles in chamber | Obscured imaging fields; false negative/low count. | Carefully reload a new cartridge if possible. |
| Incomplete mixing/vortexing | Clumped or uneven cell distribution; count variability. | Ensure strict adherence to 3-second vortex. |
| Incorrect volume (<100µL/chamber) | Reduced area scanned; potential underestimation. | Precisely calibrate and use fixed-volume pipettes. |
| Cartridge damage/scratches | Impaired focusing and automated scanning. | Visually inspect cartridge before loading. |
Visualization of Workflow
Diagram 1: CellSearch Sample Prep and Loading Workflow
Diagram 2: Cartridge Loading Schematic for Analysis
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for CellSearch Sample Loading
| Item (Part #) | Function in Loading Protocol |
|---|---|
| CellSearch Dilution Buffer (7900005) | Optimizes ionic strength and pH for cell stability. Prevents clumping and ensures even magnetic sedimentation within the cartridge. |
| CellSearch MagNest Cell Presentation Cartridge (6400002) | Proprietary dual-chambered device with integrated magnetic elements. Presents cells in a monolayer for high-resolution fluorescence scanning. |
| CellSearch Sample Tubes (12 x 75 mm) (9501001) | Specially designed tubes compatible with the system magnet and fluidics for consistent pellet formation and supernatant removal. |
| Fixed-Volume Pipettes (100 µL & 150 µL) | Critical for volumetric accuracy during the final resuspension and cartridge loading steps to ensure reproducibility. |
| Magnetic Cartridge Rack | Holds the MagNest cartridge securely during manual loading, preventing spillage or contamination. |
Within the broader thesis on the standardized CellSearch system protocol for circulating tumor cell (CTC) enumeration in metastatic cancer research, the CellTracks Analyzer II represents the critical analytical endpoint. This automated microscope and image analyzer executes the final steps of the workflow: immunofluorescent staining, high-resolution scanning, and digital image analysis of immunomagnetically enriched cells. Its role is to provide objective, reproducible identification and enumeration of CTCs defined as nucleated cells expressing epithelial cell adhesion molecule (EpCAM) and cytokeratins (CK) but lacking CD45. This application note details the protocols and technical considerations for operating the Analyzer II, which is fundamental to generating the quantitative data that correlates CTC counts with clinical outcomes in therapeutic development.
Key Research Reagent Solutions and Materials
The following table lists the essential consumables and reagents required for the staining and analysis phase on the CellTracks Analyzer II.
| Item Name | Function in the Workflow |
|---|---|
| CellSearch CTC Kit (e.g., for Breast, Prostate, Colorectal Cancer) | Provides all essential pre-optimized reagents: anti-EpCAM ferrofluid, staining reagents (nucleic acid dye, anti-CK-PE, anti-CD45-APC), fixation buffer, permeabilization buffer, and system fluid. |
| CellTracks Magnest | Device that holds the sample cartridge during staining, using magnets to retain magnetically labeled cells during automated wash steps on the Analyzer II. |
| CellTracks Sample Cartridge | A proprietary sample chamber where the enriched sample is placed for staining and scanning, ensuring consistent focal plane geometry. |
| CellSearch System Fluid | A buffered solution used as a diluent and for priming the fluidic system of the Analyzer II. |
| Control Cells (e.g., SKBR-3, PC-3) | Cultured tumor cell lines used for system performance verification and protocol validation prior to patient sample testing. |
Automated Staining and Scanning Protocol
This protocol assumes immunomagnetic enrichment from 7.5 mL of patient blood has been performed using the CellTracks Autoprep system. The sample is now contained within a Sample Cartridge.
Step 1: Instrument Preparation.
- Prime the CellTracks Analyzer II with System Fluid as per the manufacturer's startup procedure.
- Initialize the software and verify that all fluid lines are clear and the waste container is empty.
- Perform a system performance check using control cells if required by your laboratory's QC protocol.
Step 2: Cartridge Loading and Staining Setup.
- Place the Sample Cartridge containing the enriched cell sample into the CellTracks Magnest.
- Load the appropriate CellSearch CTC Kit staining reagents into the designated, temperature-controlled reagent slots on the Analyzer II. A standard kit includes:
- Reagent 1: Fixation Buffer.
- Reagent 2: Permeabilization Buffer.
- Reagent 3: Staining Mixture containing anti-CK-PE (epithelial marker) and anti-CD45-APC (leukocyte marker).
- Reagent 4: Nucleic Acid Dye (DAPI, to identify nucleated cells).
Step 3: Automated Staining and Scanning Execution.
- Place the Magnest with the cartridge into the Analyzer II.
- Select the appropriate assay protocol (e.g., "CTC Breast Cancer") on the touchscreen interface and start the run.
- The instrument automatically executes the following sequence:
- Fixation: Addition of Fixation Buffer to permeabilize and stabilize cells.
- Permeabilization: Addition of Permeabilization Buffer to allow intracellular antibody access.
- Immunofluorescent Staining: Sequential addition of the Nucleic Acid Dye and the antibody Staining Mixture, with automated incubation and wash steps between additions.
- Scanning: The cartridge is automatically transferred to the scanning stage. The microscope scans the entire surface of the sample cartridge in four fluorescence channels:
- DAPI (excitation 365 nm, emission 445-480 nm): Identifies nucleated cells.
- PE (excitation 535 nm, emission 580-595 nm): Detects cytokeratin (CK) expression.
- APC (excitation 635 nm, emission 660-700 nm): Detects CD45 expression.
- FITC (optional, excitation 480 nm, emission 505-545 nm): Can be used for an additional marker.
Image Analysis and CTC Identification Criteria
Post-scanning, proprietary CellTracks Analyzer II software analyzes all captured events based on predefined, FDA-cleared algorithms. The identification criteria are summarized in the table below.
| Parameter | CTC Definition | Leukocyte Definition | Debris/Artifact |
|---|---|---|---|
| Nucleus (DAPI) | Positive (intact, round/oval) | Positive (intact, may be lobed) | Negative or irregular |
| Cytokeratin (CK-PE) | Positive (fluorescence >= predefined threshold) | Negative | Negative |
| CD45 (CD45-APC) | Negative | Positive (fluorescence >= predefined threshold) | Negative |
| Morphology | Intact cellular structure, size >4 µm | Intact cellular structure | Does not meet cell criteria |
| Image Presentation | Composite image displays DAPI (blue), CK-PE (green), CD45-APC (red). | Composite image shows DAPI (blue) and CD45-APC (red). | - |
The software presents galleries of images that meet the CTC criteria for final operator review. The user confirms or rejects each event based on visual verification of the objective criteria. The final CTC count is reported as the number of CTCs per 7.5 mL of blood.
Data Output and Quality Control Metrics
Key quantitative outputs from the Analyzer II run should be recorded for quality assurance.
| QC Metric | Typical Acceptable Range / Value | Purpose |
|---|---|---|
| Total Number of Scanned Events | Instrument/assay dependent | Indicates scan completeness. |
| Number of Images Presented in CTC Gallery | Patient sample dependent | Raw count requiring verification. |
| Number of Verified CTCs | Final result (e.g., <5, >=5) | Primary clinical/research endpoint. |
| Control Cell Recovery (from QC runs) | Typically >70% (lab-specific) | Validates entire enrichment/staining/scanning process. |
| Background Fluorescence Levels | Below software threshold | Confirms proper washing and reagent specificity. |
Title: CellTracks Analyzer II Workflow Overview
Title: CTC Identification Logic on Analyzer II
Title: Analyzer II Multi-Channel Imaging Process
Within the broader thesis on the CellSearch system protocol for metastatic cancer research, the manual review and enumeration of Circulating Tumor Cells (CTCs) is the critical final step. This process validates the automated immuno-magnetic selection and staining, translating fluorescent signals into a clinically and scientifically meaningful count. The accuracy of this manual step is paramount, as it directly impacts patient stratification, prognosis, and therapy response assessment in clinical trials and drug development.
CTC Identification and Classification Criteria
The CellSearch system enriches cells expressing the Epithelial Cell Adhesion Molecule (EpCAM) and stains them with fluorescent reagents. A CTC is definitively identified during manual review based on the following consensus criteria:
| Criterion | Requirement | Rationale |
|---|---|---|
| DAPI (4',6-diamidino-2-phenylindole) | Positive nuclear stain. | Confirms the presence of an intact nucleated cell. |
| Cytokeratins (CK 8, 18, 19) | Positive cytoplasmic stain (PE; phycoerythrin). | Identifies epithelial origin, a hallmark of carcinoma cells. |
| CD45 (Leukocyte Common Antigen) | Negative membrane stain (APC; allophycocyanin). | Excludes leukocytes (white blood cells), the primary source of contamination. |
| Morphology | Intact cell, round to oval, with a visible nucleus. Size typically >4µm. | Distinguishes intact cells from apoptotic bodies or cellular debris. |
Classification of Observed Events: Manual reviewers classify every event in the designated analysis area of the cartridge. The primary classifications are summarized below.
| Cell Type | DAPI | Cytokeratin (CK) | CD45 | Morphology |
|---|---|---|---|---|
| CTC | Positive | Positive (clear ring) | Negative | Intact, nucleated cell. |
| Leukocyte | Positive | Negative | Positive | Often smaller, irregular shape. |
| Apoptotic CTC / Debris | Weak/Negative | Positive (punctate/diffuse) | Negative | Fragmented, no intact nucleus. |
| Artifact | Variable | Variable | Variable | Non-cellular, out-of-focus. |
Detailed Manual Review Protocol
Principle: A trained technician or scientist reviews all fluorescent images captured by the CellTracks Analyzer II to enumerate CTCs according to the defined criteria.
Materials & Equipment:
- CellTracks Analyzer II or Review Station.
- CellSearch CTC Kit (including staining reagents, capture ferrofluid, etc.).
- High-resolution monitor.
- Validated software for image display (CellTracks Software).
- Calibration slides.
Procedure:
- System Calibration & Login: Ensure the review station is calibrated using appropriate fluorescent beads. Log into the CellTracks software with reviewer credentials.
- Cartridge Loading: Physically load the processed CellSearch cartridge into the Analyzer or open the corresponding digital case file.
- Systematic Image Review: The software presents galleries of images. The reviewer systematically examines each event containing a DAPI-positive object.
- First Filter: Check for DAPI positivity to confirm a nucleated event.
- Second Filter: Assess CD45 (APC) channel. A bright ring-like stain indicates a leukocyte; mark as such.
- Third Filter: Assess Cytokeratin (PE) channel. A bright, circumferential, ring-like cytoplasmic stain is required for CTC classification.
- Final Synthesis: Overlay channels. A valid CTC must be DAPI+, CK+, CD45-, and morphologically intact.
- Enumeration & Classification: For each event, the reviewer assigns a classification (CTC, Leukocyte, Debris, etc.) via mouse click. The software tallies counts automatically.
- Quality Control: A second reviewer should re-evaluate a subset of cases (e.g., 10% or all positive cases) for verification, especially in a research setting. Discrepancies are resolved by consensus or by a senior reviewer.
- Data Export: Final CTC count (per 7.5 mL of blood) and associated images are exported for statistical analysis.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in CTC Analysis |
|---|---|
| CellSearch CTC Kit | Integrated kit containing all necessary reagents for sample preparation, immunomagnetic enrichment (EpCAM ferrofluid), and staining (DAPI, anti-CK-PE, anti-CD45-APC). |
| CellSave Preservative Tubes | Blood collection tubes containing a cellular preservative. They maintain cell integrity and prevent epitope degradation for up to 96 hours post-draw, critical for multi-site trials. |
| Control Materials (CellTracks Control Kit) | Contains stabilized human cells with known characteristics. Used to validate instrument performance, reagent integrity, and the entire assay workflow. |
| ICC/IF Validation Antibody Panels | Additional antibodies (e.g., for HER2, AR, PD-L1) for subsequent immunocytochemical/immunofluorescence staining on CellSearch cartridges to phenotype enumerated CTCs. |
| Next-Generation Sequencing (NGS) Kits | For genomic analysis of CTCs recovered after CellSearch enumeration (requires protocol modification for cell recovery), enabling mutation profiling. |
Visualization of Workflows and Pathways
Diagram 1: CellSearch CTC Analysis Workflow
Diagram 2: CTC Identification Logic Pathway
Diagram 3: CTC Phenotype & Downstream Analysis Pathways
The enumeration of Circulating Tumor Cells (CTCs) using the CellSearch system provides a prognostic and potentially predictive biomarker in metastatic cancer. Reporting these counts and interpreting them against established clinical cut-off values is critical for standardizing research and informing therapeutic decisions. This protocol is framed within a thesis on optimizing CellSearch methodology for translational research in metastatic breast, prostate, and colorectal cancers.
Key Clinical Cut-Off Values and Prognostic Data
The table below summarizes established prognostic cut-offs from pivotal clinical studies.
Table 1: Established Clinical Cut-Off Values for CTC Enumeration in Metastatic Cancers
| Cancer Type (Metastatic) | Prognostic Cut-Off (CTCs/7.5 mL) | Associated Clinical Outcome | Key Supporting Study |
|---|---|---|---|
| Breast | ≥5 | Significantly shorter Median Progression-Free Survival (PFS) and Overall Survival (OS) | Cristofanilli et al., NEJM, 2004 |
| Prostate (Castration-Resistant) | ≥5 | Shorter OS and time to oncologic events | de Bono et al., Clin Cancer Res, 2008 |
| Colorectal | ≥3 | Shorter PFS and OS | Cohen et al., JCO, 2008 |
| General Carcinoma* | ≥5 | Stratifies patients into favorable/unfavorable prognostic groups | Multiple validations |
*Applies to metastatic breast, prostate, colorectal, and other epithelial cancers.
Table 2: Categorization of CTC Counts and Clinical Interpretation
| CTC Count (per 7.5 mL) | Prognostic Category | Typical Clinical Research Interpretation |
|---|---|---|
| 0 | Favorable | Low risk of progression; potential for better treatment response. |
| 1 - 4 | Intermediate | Requires careful monitoring; consider integration with other biomarkers. |
| ≥5 | Unfavorable | High risk of disease progression and shorter survival; candidate for aggressive or novel therapy. |
Detailed Protocol: CellSearch CTC Enumeration and Reporting
Materials and Pre-Analysis Preparation
- CellSearch System: Includes CellTracks AutoPrep system, CellTracks Magnest, and CellTracks Analyzer II.
- CellSearch CTC Kit: Contains ferrofluid nanoparticles (anti-EpCAM), staining reagents (anti-CK-PE, anti-CD45-APC, DAPI), preservative, and buffer.
- Sample: 7.5 mL of peripheral blood drawn into CellSave Preservative Tubes.
- Quality Control: Process control materials (e.g., control blood with spiked tumor cells) must be run with each batch.
Step-by-Step Workflow Protocol
- Sample Acceptance & Registration: Verify blood volume (7.5 mL), tube type (CellSave), and time from draw (<96 hours at room temp). Log sample into the CellTracks database.
- Automated Immunomagnetic Enrichment (CellTracks AutoPrep): a. Load samples and the CellSearch CTC Kit. b. The system immunomagnetically enriches epithelial cells using anti-EpCAM ferrofluid. c. Cells are stained with fluorescent reagents: phycoerythrin (PE)-conjugated anti-cytokeratin (CK) to identify epithelial cells, allophycocyanin (APC)-conjugated anti-CD45 to identify leukocytes, and 4',6-diamidino-2-phenylindole (DAPI) to stain nucleic acid. d. Enriched and stained cells are automatically dispensed into a cartridge (Magnest).
- Magnetic Cartridge Assembly: The cartridge is placed in the CellTracks Magnest, which positions the cells for analysis.
- Automated Digital Microscopy & Image Acquisition (CellTracks Analyzer II): The system scans the entire cartridge surface, capturing digital images from four fluorescent channels (PE, APC, DAPI, and FITC).
- CTC Identification & Enumeration: a. An algorithm presents galleries of objects that are CK-PE positive, DAPI positive, and CD45-APC negative. b. A certified operator reviews each potential CTC image based on stringent criteria: * Positive CTC: Round/oval morphology, CK-PE (+), DAPI (+), CD45-APC (-), with cytoplasmic CK staining. * Negative (Leukocyte): CK-PE (-), DAPI (+/-), CD45-APC (+). * Negative (Debris/Artifact): Irregular shape, CK-PE staining artifacts, or incomplete DAPI staining. c. The final CTC count is manually confirmed and recorded.
Mandatory Data Reporting Standards
For publication and internal documentation, report:
- Absolute CTC Count: Number of CTCs per 7.5 mL of blood.
- Categorical Result: Classify as "<5 CTCs" or "≥5 CTCs" (or relevant study-specific cut-off).
- Patient/Sample Identifier
- Assay Date & Lot Numbers for reagents.
- Operator ID who performed the review.
- Any deviations from the standard protocol.
Signaling Pathways Related to CTC Biology
The survival and proliferation of CTCs involve key pathways that are targets for novel therapies.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for CTC Research via CellSearch
| Item | Function in CTC Workflow | Key Consideration |
|---|---|---|
| CellSave Preservative Tubes | Stabilizes blood cells and prevents EpCAM degradation for up to 96 hours. | Must be used for collection; strict adherence to hold time is critical. |
| CellSearch CTC Kits (Cancer-specific) | Provides all reagents for immunomagnetic enrichment (anti-EpCAM) and fluorescent staining (CK, CD45, DAPI). | Kit type must match cancer origin (e.g., breast, prostate, general carcinoma). |
| CellSearch Control Kits | Contains stabilized tumor cells for spiking into control blood. Used for process validation and monitoring operator performance. | Essential for daily QC and assay qualification. |
| Pipette Tips & Reagent Troughs (CellSearch Certified) | Disposable supplies designed for use with the AutoPrep system. | Non-certified supplies may cause instrument errors. |
| Calibration Beads (CellSearch) | Used for periodic instrument calibration of the Analyzer II fluorescence optics. | Required for maintaining quantitative imaging consistency. |
| Independent Anti-EpCAM Antibodies (for research) | Used for complementary CTC capture experiments (e.g., microfluidic devices) to compare with CellSearch yield. | Different EpCAM clones or affinities may capture heterogeneous CTC subsets. |
| RNA/DNA Stabilization Reagents (e.g., PAXgene) | For preserving nucleic acids from isolated CTCs for downstream molecular characterization (post-CellSearch). | Enables genomic, transcriptomic, or single-cell analysis of enumerated CTCs. |
CellSearch Troubleshooting Guide: Solving Common Problems and Optimizing Assay Performance
The reliable enumeration of Circulating Tumor Cells (CTCs) using the CellSearch system is a critical prognostic and predictive tool in metastatic cancer research. However, the analytical integrity of this liquid biopsy approach is profoundly vulnerable to pre-analytical errors. Hemolysis, clotting, and delays in sample processing introduce artifacts and biological degradation that directly compromise the accuracy, precision, and clinical relevance of CTC counts. This document provides detailed application notes and protocols to mitigate these variables, framed within a broader thesis aimed at standardizing the CellSearch workflow for robust, reproducible data in clinical trials and drug development.
Table 1: Impact of Hemolysis, Clotting, and Delay on CellSearch Outcomes
| Pre-Analytical Variable | Key Effect on Sample | Impact on CellSearch CTC Enumeration | Reported Reduction in CTC Recovery |
|---|---|---|---|
| Hemolysis | Release of hemoglobin and intracellular components; generates debris. | Increased background fluorescence; non-specific antibody binding; potential EpCAM antigen degradation. | 15-40% (Severe hemolysis; H-index >100) |
| Clotting / Microclots | Physical entrapment of CTCs in fibrin mesh; clogging of system capillaries. | Complete loss of entrapped CTCs; assay failure due to instrument clogging; invalid results. | Up to 100% for entrapped cells |
| Delayed Processing | Leukocyte degradation; CTC apoptosis and loss of membrane integrity (EpCAM, CK). | Reduced immunofluorescence signal; increased false negatives; elevated background from lysed WBCs. | 10-25% per 24h at RT; >50% after 72h at RT (varies by cancer type) |
| Incorrect Draw Volume | Underfilling alters blood-to-preservative ratio (CellSave tube). Overfilling risks inadequate mixing and clotting. | Suboptimal cell preservation; increased clotting risk; off-label conditions affecting FDA-cleared protocol performance. | Variable; can invalidate test |
| Temperature Excursion | Sample storage outside 4-27°C range for CellSave tubes. | Accelerated cell degradation or preservative toxicity. | Not quantified but considered a major protocol deviation. |
Detailed Experimental Protocols for Mitigation
Protocol 3.1: Standardized Blood Collection & Handling for CellSearch
Objective: To ensure consistent pre-analytical conditions for CTC enumeration. Materials: CellSave Preservative Blood Collection Tubes (10mL), 21-gauge or larger butterfly needle, tourniquet, alcohol swabs, tube mixer. Procedure:
- Patient Identification & Preparation: Confirm patient identity. Minimize patient stress to reduce hemodilution or physiologically induced changes.
- Venipuncture: Use a large, clean vein. Disinfect site. Do not draw from lines unless absolutely necessary (high hemolysis/clot risk). Discard first 2-3 mL if using a standard needle to avoid tissue contamination.
- Tube Filling: Draw blood slowly and smoothly to avoid mechanical hemolysis. Fill the CellSave tube to exactly 10 mL (±0.5 mL). The tube contains EDTA and a cellular preservative; under/overfilling alters preservation chemistry.
- Immediate Mixing: Invert tube gently 10 times immediately after draw to ensure complete mixing with preservative.
- Labeling & Documentation: Note exact draw time on tube.
- Storage & Transport: Store tubes upright at room temperature (15-27°C). Do not refrigerate or freeze. Process within 96 hours of draw (72 hours is the strongly recommended stability window).
Protocol 3.2: Visual Inspection and Sample Rejection Criteria
Objective: To identify samples compromised by hemolysis or clotting prior to processing. Materials: Bright light source, white background. Procedure:
- Inspect for Clots: Gently invert tube twice. Examine for any visible fibrin strands, clots, or "sludging." Note: Microclots may not be visible.
- Assess Hemolysis: Hold tube against a white background. Compare plasma color to a standard hemolysis index chart.
- Acceptable: Pale yellow.
- Caution (May Proceed): Slight pink hue.
- Reject (Do Not Process): Red or cherry-red plasma. Document H-index if available.
- Document & Decision: Record observations. A visibly clotted or severely hemolyzed sample should be rejected, the clinical team notified, and a recollection requested if feasible.
Protocol 3.3: Protocol for Investigating Delay-to-Processing Effects (Stability Study)
Objective: To empirically determine CTC stability in your specific laboratory setting. Materials: Multiple CellSave tubes from consented metastatic cancer patients (research protocol), CellSearch system, data analysis software. Experimental Design:
- Baseline (T0): Process one 10mL tube from a patient within 4 hours of draw. Record CTC count.
- Delayed Time Points: From the same patient draw, process identical tubes at T=24h, 48h, 72h, and 96h stored at RT.
- Replicates: Perform for N≥10 patient samples.
- Analysis: Calculate mean % CTC recovery at each time point relative to T0. Plot decay curve. Perform statistical analysis (e.g., paired t-test) to determine the time point where significant loss occurs.
Visualized Workflows and Relationships
Title: Pre-Analytical Errors Lead to Low CTC Recovery
Title: CellSearch Pre-Analytical Quality Control Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Materials for Managing Pre-Analytical Variables
| Item | Function / Rationale | Example / Specification |
|---|---|---|
| CellSave Preservative Tube | Contains EDTA (anticoagulant) and a proprietary cell preservative to stabilize EpCAM and cytokeratin antigens on CTCs for up to 96 hours. Critical for protocol. | Menarini Silicon Biosystems, 10mL draw. CE-IVD/FDA-cleared. |
| Hemolysis Index (H-index) Standards | Provides semi-quantitative visual or spectrophotometric assessment of plasma hemoglobin to standardize sample rejection criteria. | Commercially available color charts or integrated serum indices on clinical analyzers. |
| 21-Gauge or Larger Butterfly Needle | Minimizes shear stress during blood draw, reducing mechanical hemolysis risk compared to smaller gauge needles. | Standard medical device. Avoid 23G or smaller for CellSearch draws. |
| Programmable Tube Mixer | Ensures consistent, gentle, and immediate mixing of blood with preservative post-draw, preventing microclot formation. | Lab accessory set to slow tilt-rotation, not vortex. |
| Temperature Data Logger | Monitors and documents ambient temperature during sample storage and transport to ensure compliance with the 15-27°C range. | Small USB or Bluetooth loggers placed with sample batches. |
| Alternative Preservation Tubes (Research) | For extended stability studies or remote collection. Contain stronger fixatives or RNA stabilizers. Note: Require separate validation against CellSave. | Streck Cell-Free DNA BCT, PAXgene Blood ccfDNA Tube. |
| Fibrin Clot Dissociation Agents (Research-Use Only) | Enzymatic treatment (e.g., thrombin inhibitors, fibrinolysin) to potentially recover CTCs from slightly clotted samples. Not part of standard CellSearch protocol. | Research-grade enzymes. Validation required for each cancer type. |
Troubleshooting Low Cell Yield or Poor Staining Quality
Within the broader thesis on the CellSearch system for circulating tumor cell (CTC) enumeration in metastatic cancer research, consistent cell yield and optimal staining quality are paramount for reliable data. Low yields can compromise statistical power, while poor staining confounds accurate CTC identification. This application note details common failure points and provides protocols for systematic troubleshooting.
The following table summarizes key variables affecting performance, based on current literature and system specifications.
Table 1: Primary Factors Impacting CellSearch Assay Performance
| Factor | Potential Impact on Yield | Potential Impact on Staining | Typical Optimal Range/Standard |
|---|---|---|---|
| Blood Collection & Handling | High | Moderate | CellSave Preservative Tube; Processed within 96h (72h recommended); Gentle inversion 8x |
| Sample Volume | Directly Proportional | N/A | 7.5 mL; precise volumetric draw critical |
| Centrifugation | High (Cell Loss) | Moderate (Cell Integrity) | System-defined; Ensure proper deceleration setting |
| Magnetic Separation | High (Inefficient Capture) | Low | Ensure proper pipettor magnet engagement timing |
| Reagent Quality & Storage | Moderate | High | Validated lots; 2-8°C storage; Protect fluorescent probes from light |
| Instrument Calibration | Moderate | High | Regular system performance checks (SPC) with control cells |
| Fixation & Permeabilization | Low | High (Epitope Masking) | Strict adherence to incubation times and temperatures |
| Endogenous Factors (Patient) | High (True Biology) | Low | Disease burden, treatment effects, epithelial-mesenchymal transition (EMT) |
Detailed Experimental Protocols
Protocol 1: Systematic Verification of Pre-Analytical Variables
Objective: To isolate and confirm issues arising from sample procurement and handling.
- Materials: CellSave Preservative Tubes, timer, calibrated pipettes.
- Method: a. Draw Verification: Confirm blood draw is exactly 7.5 mL into CellSave tube. Under-filling reduces yield; over-filling compromises preservation. b. Mixing: Invert tube gently 8-10 times immediately after draw to mix preservative. c. Time Log: Record exact time of collection. Prioritize processing for samples >72h old. d. Shipping Temp: Verify samples were maintained at ambient temperature (15-25°C); do not refrigerate or freeze.
- Expected Outcome: Eliminates pre-analytical error as a root cause.
Protocol 2: Staining Quality Control & Titration
Objective: To diagnose and resolve issues with fluorescent signal intensity or specificity.
- Materials: CellSearch CXC Kit, fluorescent calibration beads, light-blocking containers.
- Method: a. Reagent Inspection: Visually check reagents for precipitates or discoloration. Ensure all fluorescent antibodies (CK-PE, DAPI, CD45-APC) are stored in dark. b. System Performance Check (SPC): Run the commercial SPC cartridge. If control cell signals are low, perform instrument optical path calibration. c. Epitope Retrieval Test (if applicable): For suspected epitope masking due to fixation, test an additional, validated permeabilization step (e.g., 0.1% Triton X-100 for 5 mins post-fixation) on a test sample. d. Negative Control: Always include a sample stained with isotype control antibodies to assess non-specific binding.
- Expected Outcome: Identifies whether poor staining is due to reagent, instrument, or sample-specific factors.
Visualizing the Troubleshooting Workflow
Troubleshooting Low Yield and Poor Staining
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for CellSearch Troubleshooting
| Item | Function in Troubleshooting |
|---|---|
| CellSave Preservative Tubes | Standardized blood collection for CTC stability; primary variable to control. |
| CellSearch System Performance Check Kit | Validates instrument fluidics, optics, and thermal systems using control cell lines. |
| Fluorescent Calibration Beads | Independent verification of detector sensitivity and laser alignment. |
| Validated Isotype Control Antibodies | Distinguish specific from non-specific antibody binding in staining assays. |
| Alternative Fixation/Permeabilization Buffer | Tests epitope accessibility if standard protocol masks target antigens. |
| Reference Samples (High/No CTC) | Positive and negative process controls for the entire workflow. |
| Precision Calibrated Pipettes | Ensures accurate reagent delivery during the multi-step assay. |
1. Introduction Within the established thesis on the CellSearch system for circulating tumor cell (CTC) enumeration in metastatic cancer research, a significant limitation arises in cancers with low or heterogeneous expression of EpCAM. This application note details strategies and protocols to optimize detection and analysis of CTCs in these challenging cases, ensuring comprehensive liquid biopsy profiling.
2. Strategies and Complementary Approaches While CellSearch remains the FDA-cleared standard, its reliance on epithelial cell adhesion molecule (EpCAM) capture necessitates supplementary methods for certain cancer types.
Table 1: Comparison of CTC Enrichment Strategies for Low-EpCAM Cancers
| Strategy | Principle | Target | Advantages | Limitations |
|---|---|---|---|---|
| Negative Selection (Depletion) | Remove CD45+ leukocytes and other hematopoietic cells. | All nucleated, non-hematopoietic cells. | EpCAM-independent; captures epithelial, mesenchymal, and EMT-ing CTCs. | Low purity; requires high downstream resolution. |
| Size-Based Filtration | Isolate cells larger than pore size (e.g., 6.5-8 µm). | Large, intact cells. | Label-free; preserves cell viability. | Can miss small CTCs; clogging; leukocyte contamination. |
| Density Gradient Centrifugation | Separate cells based on buoyant density. | Mononuclear cell layer. | Simple, cost-effective; enriches for viable cells. | Low specificity and purity; CTC loss. |
| Combined Positive & Negative Immunomagnetic | Positive select for EpCAMand negative deplete for CD45. | CTCs with variable EpCAM. | Improves specificity over negative alone. | Complex protocol; potential loss. |
| Microfluidic & Chip-Based | Use affinity (alternative antibodies) or biophysical properties. | EpCAM, other markers (EGFR, HER2) or size/deformability. | High capture efficiency; can use whole blood. | Platform-dependent; not yet standardized. |
3. Detailed Application Protocols
Protocol 3.1: Combined Negative Depletion and Positive Picking for CTC Enrichment Objective: To enrich for CTCs from whole blood using a two-step immunomagnetic protocol, minimizing EpCAM bias. Materials: Anti-CD45 magnetic beads (e.g., Dynabeads), anti-EpCAM ferrofluid (CellSearch), magnet, PBS/BSA/EDTA buffer, rotator. Workflow:
- Leukocyte Depletion: Incubate 7.5 mL of anti-coagulated blood with anti-CD45 beads (per manufacturer's ratio) for 30 min at 4°C on a rotator.
- Place tube on a magnet for 2-5 min. Carefully transfer the supernatant (containing unbound cells) to a new tube. Discard the bead-bound CD45+ fraction.
- EpCAM-Dependent Capture: Add CellSearch anti-EpCAM ferrofluid to the supernatant. Incubate for 10 min at room temperature.
- Place tube on the magnet. Discard supernatant. Resuspend the magnetically captured cell fraction in PBS/BSA/EDTA for downstream staining or analysis on the CellSearch system.
Protocol 3.2: Downstream Immunofluorescence & Characterization of Enriched CTCs Objective: To identify and phenotype CTCs post-enrichment using a multi-marker panel. Materials: Cytospin centrifuge, slides, paraformaldehyde (4%), Triton X-100 (0.1%), blocking buffer (3% BSA), primary and secondary antibodies, DAPI. Workflow:
- Cell Fixation & Permeabilization: Cytospin enriched cells onto slides. Fix with 4% PFA for 15 min. Permeabilize with 0.1% Triton X-100 for 10 min.
- Blocking: Apply 3% BSA for 30 min to reduce non-specific binding.
- Antibody Staining: Incubate with primary antibody cocktail (e.g., anti-CK8,18,19, anti-HER2, anti-Vimentin) for 60 min at RT or overnight at 4°C.
- Wash 3x with PBS. Incubate with fluorophore-conjugated secondary antibodies (and/or fluorescently labeled anti-CD45) for 45 min in the dark.
- Counterstaining & Mounting: Stain nuclei with DAPI for 5 min. Wash, mount, and seal.
- Imaging & Analysis: Use a fluorescent microscope or scanning system. Define CTCs as DAPI+, CK+, CD45-. Note CK-low/negative, vimentin-positive cells.
4. Visualizing Strategies and Pathways
Title: Multimodal CTC Enrichment Workflow for Low-EpCAM Cancers
Title: EMT Signaling Leads to EpCAM-Low CTC Phenotype
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Optimized CTC Studies
| Reagent/Material | Function | Example/Note |
|---|---|---|
| Anti-EpCAM Ferrofluid (CellSearch) | Immunomagnetic positive selection of epithelial CTCs. | Gold standard; used in primary or combined protocols. |
| Anti-CD45 Magnetic Beads | Negative selection by depletion of leukocytes. | Enables EpCAM-independent pre-enrichment. |
| Cytokeratin (CK) Antibody Cocktail | Pan-epithelial marker for CTC identification. | CK8, 18, 19; detect epithelial CTCs. |
| Anti-Vimentin Antibody | Mesenchymal marker for EMT-ing CTC detection. | Critical for phenotyping low-EpCAM CTCs. |
| Anti-CD45 Antibody (Fluor-conj.) | Leukocyte exclusion marker in immunofluorescence. | Differentiates CTCs (CD45-) from WBCs. |
| Nucleic Acid Preservation Buffer | Stabilizes RNA/DNA for molecular analysis post-enrichment. | Enables single-CTC genomics/transcriptomics. |
| Microfluidic CTC Capture Chip | EpCAM or size-based capture from whole blood. | Platform-dependent (e.g., Herringbone, NanoVelcro). |
| DAPI (4',6-diamidino-2-phenylindole) | Nuclear counterstain for cell viability/identification. | Defines intact nucleated cells. |
Mitigating Operator Error and Ensuring Inter-Operator Reproducibility
Within the broader thesis on the CellSearch system protocol for circulating tumor cell (CTC) enumeration in metastatic cancer research, the precision and accuracy of results are paramount. This protocol hinges on consistent operator performance. Operator-dependent variability in sample handling, instrument operation, and image interpretation is a documented source of error, potentially impacting clinical trial outcomes and longitudinal patient monitoring. These Application Notes provide a detailed framework for standardizing the CellSearch workflow, implementing rigorous quality control (QC) metrics, and establishing training programs to minimize operator-induced variance and ensure inter-operator reproducibility.
Critical Operator-Dependent Variables in CellSearch CTC Enumeration
The following table summarizes key procedural steps where operator error can significantly impact results, along with proposed mitigation strategies.
Table 1: Critical Variables and Mitigation Strategies
| Procedural Step | Potential Operator Error | Impact on CTC Enumeration | Mitigation Strategy |
|---|---|---|---|
| Blood Sample Handling | Incorrect fill volume; improper mixing; delayed processing. | CTC loss or degradation; clot formation. | Use of precisely calibrated collection tubes; strict adherence to <96h processing window; trained on vortex mixing protocol. |
| Sample Preparation (AutoPrep) | Reagent loading errors; incorrect cartridge placement; mislabeling. | Assay failure; sample cross-contamination. | Use of barcode scanning for reagent/sample ID; checklist for cartridge alignment; two-operator verification for critical steps. |
| Immunomagnetic Enrichment & Staining | Deviation from protocol timing; improper pipetting technique. | Inconsistent epithelial cell capture; non-specific staining. | Calibrated, automated pipettes; timed protocol with audible alerts; use of control samples. |
| Cartridge Loading (MagNest) | Improper seating of cartridge; bubbles in sample chamber. | Poor image quality; cell loss during scanning. | Standardized loading fixture; visual inspection checklist. |
| Image Acquisition & Analysis | Inconsistent focus adjustment; subjective fluorescence thresholding. | Variable detection sensitivity; false positives/negatives. | Automated focus algorithms; standardized software settings; mandatory review of predefined gallery fields. |
| CTC Identification & Enumeration | Subjective interpretation of phenotype (CK+/DAPI+/CD45-). | High inter-operator variability in final count. | Dual-blinded review by two certified operators; use of consensus guidelines with reference images. |
Protocol for Standardized Operator Training and Certification
Objective: To ensure all operators achieve a baseline competency, reducing subjective variance in the final interpretation step. Materials: CellSearch System, CellSearch Circulating Tumor Cell Kit, control blood samples (healthy donor spiked with cultured tumor cells, e.g., SKBR-3 or PC-3), CellTracks Analyzer II.
Procedure:
- Theoretical Training: Complete manufacturer's training modules on system principles, standard operating procedure (SOP), and pre-analytical variables.
- Hands-On Proficiency: Under supervision, process a minimum of 10 control samples (with known, pre-defined CTC spike-in counts ranging from 0 to >100 cells).
- Image Analysis Certification: a. Each operator independently analyzes a standardized set of 50 previously acquired patient sample galleries. b. Results are compared against a consensus "gold standard" count established by three senior, certified technicians. c. Certification Criteria: Operator must achieve ≥95% concordance with the gold standard count and 100% correct classification of control samples (positive vs. negative) to pass.
- Recertification: Mandatory every 6 months using a new standardized test set.
Protocol for Inter-Operator Reproducibility Assessment
Objective: To quantitatively measure and monitor variability between operators within a lab. Experimental Design: A reproducibility study using replicated samples.
Procedure:
- Sample Preparation: Aliquot a large volume of control blood (spiked with a mid-range CTC count, e.g., 20-50 cells) into 10 identical CellSave tubes.
- Sample Processing: Three different certified operators independently process 3 tubes each (randomly assigned), with one tube reserved as a spare.
- Blinded Analysis: All resulting image galleries are coded and randomized. Each operator then analyzes all galleries (including their own and others') in a blinded fashion.
- Data Analysis: Calculate the coefficient of variation (CV) for the final CTC counts across all operators and samples. Use statistical tests (e.g., ANOVA) to determine if significant differences exist between operators. Acceptance Criterion: The inter-operator CV for control samples should be <20%.
Table 2: Example Reproducibility Study Data
| Operator | Sample 1 CTC Count | Sample 2 CTC Count | Sample 3 CTC Count | Mean Count (per operator) | Std Dev |
|---|---|---|---|---|---|
| Operator A | 34 | 29 | 31 | 31.3 | 2.5 |
| Operator B | 28 | 32 | 30 | 30.0 | 2.0 |
| Operator C | 36 | 31 | 29 | 32.0 | 3.6 |
| Overall Mean: 31.1 | Overall Std Dev: 2.8 | Inter-Operator CV: 9.0% |
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for Standardized CellSearch Operation
| Item | Function in Mitigating Error |
|---|---|
| CellSave Preservative Tubes | Maintains cell integrity for up to 96 hours, standardizing pre-analytical delay variable. |
| CellSearch CTC Kit (Epithelial) | Standardized, pre-packaged reagent kit containing ferrofluid, staining reagents, and buffers to ensure lot-to-lot consistency. |
| CellSearch Control Kit | Contains lyophilized control cells for verifying system and assay performance daily, providing a benchmark for operator technique. |
| CellTracks AutoPrep System | Automates the precise fluid handling, incubation, and washing steps, minimizing manual pipetting errors. |
| Calibrated Precision Pipettes | Used for loading reagents onto the AutoPrep; regular calibration is essential for volumetric accuracy. |
| MagNest Cell Presentation Device | Standardizes the magnetic immersion of cells into a single focal plane for consistent imaging. |
| CellTracks Analyzer II | Semi-automated fluorescence microscope with standardized filter sets and digital imaging protocol. |
| Validated Reference Image Gallery | A curated set of example images (CK+/DAPI+/CD45- CTCs, CK-/DAPI+/CD45+ leukocytes, artifacts) for operator training and ongoing proficiency testing. |
Visualized Workflows and Relationships
CellSearch Workflow with Integrated Operator QC
Error Mitigation Strategy Logic Map
1. Introduction and Thesis Context Within the broader thesis on implementing a standardized CellSearch system protocol for circulating tumor cell (CTC) enumeration in metastatic cancer research, rigorous instrument maintenance and quality control (QC) are non-negotiable pillars. The clinical and research validity of CTC counts—used for prognostic assessment, therapy monitoring, and drug development endpoints—hinges on the consistent performance of the CellSearch platform. This document outlines detailed application notes and protocols for running controls and system suitability tests to ensure data integrity and inter-laboratory reproducibility.
2. The Importance of Controls in CTC Enumeration The CellSearch system automates the immunomagnetic enrichment and fluorescent staining of EpCAM-positive CTCs from whole blood. Variability can be introduced at multiple points: instrument fluidics, magnet performance, optical alignment, and reagent stability. Systematic QC using defined controls mitigates this, distinguishing true biological variation from technical artifact, a critical factor for longitudinal studies in metastatic breast, prostate, and colorectal cancer.
3. Key QC Metrics and Quantitative Data Summary The following tables summarize target values and frequencies for key QC operations. Values are derived from manufacturer specifications and current laboratory standards.
Table 1: Daily and Procedural Control Requirements
| Control Type | Material | Target Value/Criteria | Frequency | Purpose |
|---|---|---|---|---|
| System Suitability Test (SST) | CELLTRACKS CK Performance Reagents | Recovery ≥7 CTCs per vial (from 4.0-7.5 mL) | With each patient batch | Verifies overall system performance (immunomagnetic capture, staining, analysis). |
| Process Control | Pre-stained, fixed cells (e.g., SKBR-3 line) | Pre-defined recovery & fluorescence intensity ranges. | With each patient batch | Controls for sample preparation, staining, and instrument setup. |
| Negative Control | Blood from healthy donor or CELLTRACKS Control Blood | CTC count = 0 | With each patient batch | Establishes background and specificity threshold. |
| Buffer/Reagent Check | PBS/BSA Buffer | Visual inspection for clarity, precipitation. | Daily, before run | Prevents fluidic clogs and non-specific binding. |
Table 2: Periodic Instrument Performance Checks
| Check | Parameter | Acceptance Range | Frequency | Purpose |
|---|---|---|---|---|
| Optical Alignment | Fluorescence Intensity (FI) of calibration beads | FI CV < 5% for each channel (DAPI, PE, APC, Cy7). | Weekly & after maintenance | Ensures sensitivity and specificity of CTC identification. |
| Pipetting Accuracy | Volume verification via gravimetric analysis. | Deviation < 2% for critical volumes (e.g., 7.5 mL blood draw). | Monthly | Guarantees consistent sample and reagent volumes. |
| Magnet Performance | Magnetic particle capture efficiency. | Visual verification of defined bead pattern. | Daily | Critical for EpCAM+ cell enrichment efficiency. |
| Environmental Logger | Temperature in reagent storage. | 2-8°C for reagents; 15-30°C for instrument. | Continuous monitoring | Ensures reagent stability and instrument function. |
4. Detailed Experimental Protocols
Protocol 4.1: Weekly Optical Alignment and Calibration Objective: To verify and adjust the fluorescence detection optics of the CellTracks Analyzer II. Materials: CELLTRACKS Calibration Beads, CELLTRACKS Analyzer II, computer with CellTracks Software.
- Power on the Analyzer and launch the software. Select 'Calibration' mode.
- Vortex the vial of calibration beads for 15 seconds. Pipette 10 µL onto a clean, dry CELLTRACKS cartridge and seal with a MagNest cell presentation lid.
- Load the cartridge onto the Analyzer stage. Initiate the calibration scan via software.
- The software automatically scans beads in all four fluorescent channels (DAPI, PE, APC, Cy7). It reports the Mean Fluorescence Intensity (MFI) and Coefficient of Variation (CV%) for each channel.
- Acceptance Criteria: CV% for each channel must be <5%. If any CV exceeds 5%, repeat the calibration. A persistent failure indicates a need for service. Document all results in the instrument log.
Protocol 4.2: System Suitability Test (SST) with Patient Samples Objective: To confirm the entire CellSearch system is performing within specification prior to processing patient samples. Materials: CELLTRACKS CK Performance Reagent (contains pre-defined number of cultured tumor cells), CELLTRACKS AutoPrep System, relevant CellSearch CXC Kit (e.g., for EpCAM), CellTracks Analyzer II.
- Remove the CK Performance Reagent vial from 2-8°C storage. Allow it to equilibrate to room temperature (approx. 30 min).
- Treat the vial as a "patient sample." Place it in the designated rack position on the AutoPrep system.
- Load all necessary reagent kits (CXC Kit, Staining Reagents, etc.) onto the AutoPrep as per the standard workflow for patient samples.
- Run the "CELLSEARCH Profile" for the appropriate cancer type. The system will automatically spike the CK cells into the assay, process, stain, and prepare the cartridge.
- After processing, scan the cartridge on the Analyzer.
- Analysis & Acceptance: The software will identify and enumerate the recovered CK cells (positive for EpCAM, CK-PE, DAPI, and negative for CD45-APC). The result must meet the lot-specific certificate of analysis (typical range: 7-30 cells recovered). If recovery is below specification, patient sample processing must not proceed. Troubleshoot instrument and reagents.
5. Visualization: QC Workflow and Failure Analysis
Title: CellSearch QC Decision Workflow
6. The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for CellSearch QC
| Item | Function in QC |
|---|---|
| CELLTRACKS CK Performance Reagent | Contains a known quantity of fixed, stained tumor cells. The primary material for the mandatory System Suitability Test (SST) to validate recovery. |
| CELLTRACKS Calibration Beads | Fluorescent beads with defined intensity across all detection channels. Used for weekly optical alignment to ensure detection sensitivity and consistency. |
| CellSearch CXC Kit (Tumor Type Specific) | Contains the immunomagnetic ferrofluid (anti-EpCAM), staining reagents (CK-PE, CD45-APC, DAPI), and buffers. The core reagent set; lot consistency is critical. |
| Process Control Cells (e.g., SKBR-3) | Cultured cell lines spiked into healthy donor blood. Serves as an independent control for user sample preparation steps outside the AutoPrep. |
| CELLTRACKS Control Blood | Prescreened healthy donor blood. Used as a negative control (CTC=0) to verify staining specificity and rule out background interference. |
| CELLTRACKS MagNest | Magnetic cartridge holder. Its consistent magnetic field strength is vital for proper cell presentation during imaging; inspect for physical damage. |
Validating CellSearch: Clinical Evidence, Technical Comparisons, and Future Directions
Application Notes
Circulating Tumor Cell (CTC) enumeration via the CellSearch system has been validated as a prognostic and predictive biomarker across multiple metastatic carcinomas. This Application Note synthesizes the pivotal studies that established the clinical utility of CTC counts, detailing their methodologies, outcomes, and implications for clinical research and therapeutic development.
Prognostic Validation in Metastatic Breast Cancer (MBC)
The seminal study by Cristofanilli et al. (2004) in the New England Journal of Medicine established the prognostic value of a CTC count ≥5 per 7.5 mL of blood in MBC. This threshold independently predicted inferior Progression-Free Survival (PFS) and Overall Survival (OS) at baseline and first follow-up. Subsequent meta-analyses have confirmed this robust association.
Predictive Utility in Metastatic Colorectal Cancer (mCRC)
The CA19-9 study by Cohen et al. (2008) in the Journal of Clinical Oncology demonstrated that patients with mCRC and <3 CTCs at baseline and follow-up had significantly better PFS and OS compared to those with ≥3 CTCs. This validated CTCs as an early marker of treatment efficacy.
Validation in Metastatic Castration-Resistant Prostate Cancer (mCRPC)
The IMMC38 trial by de Bono et al. (2008) in Clinical Cancer Research established a prognostic threshold of ≥5 CTCs in mCRPC. CTC count was a stronger predictor of OS than PSA changes, leading to its recognition as a surrogate endpoint biomarker in clinical trials.
Real-Time Treatment Monitoring
Multiple studies across cancer types have shown that changes in CTC counts after initiating therapy (often as early as 3-4 weeks) correlate with radiographic response and survival outcomes, enabling dynamic assessment of treatment benefit.
Table 1: Landmark Studies in CTC Clinical Validation
| Cancer Type (Study) | CTC Threshold (per 7.5 mL) | Key Prognostic Outcome | Hazard Ratio (HR) [95% CI] |
|---|---|---|---|
| Metastatic Breast Cancer (Cristofanilli et al., 2004) | ≥5 | Shorter Median PFS & OS | OS HR: 2.6 [1.8-3.9] |
| Metastatic Colorectal Cancer (Cohen et al., 2008) | ≥3 | Shorter Median PFS & OS | PFS HR: 3.8 [2.4-6.0]; OS HR: 5.5 [3.2-9.5] |
| Metastatic Prostate Cancer (de Bono et al., 2008) | ≥5 | Shorter Median OS | OS HR: 6.5 [3.8-11.3] |
| Metastatic Breast Cancer (SWOG S0500, 2014) | ≥5 at 1st follow-up | No OS benefit from early therapy switch | OS HR: 1.1 [0.81-1.46] |
Table 2: CTC Count as a Predictive Biomarker for Treatment Response
| Study Context | Timing of CTC Assessment | Predictive Value |
|---|---|---|
| MBC Post-Therapy Initiation | Baseline & 3-4 weeks post-treatment | CTC conversion (<5) associated with improved clinical benefit vs. non-conversion. |
| mCRC on 1st/2nd Line Therapy | Baseline & 3-8 weeks post-treatment | Favorable CTC count (<3) correlates with objective radiographic response. |
| mCRPC on various therapies | Baseline & monthly intervals | ≥5 CTCs post-treatment indicates poor response; CTC changes correlate with OS. |
Experimental Protocols
Protocol 1: CellSearch CTC Enumeration for Prognostic Stratification
Purpose: To enumerate CTCs from peripheral blood for baseline prognostic stratification in metastatic cancer. Materials: CellSave Preservative Tubes, CellSearch CTC Kit (Ferrofluid: anti-EpCAM, Staining Reagents: anti-CK-PE, anti-CD45-APC, DAPI), CellSearch AutoPrep and Analyzer Systems. Procedure:
- Blood Collection & Storage: Draw 7.5 mL blood into CellSave tube. Invert 8x to mix. Stable at room temperature for up to 96 hours.
- Sample Preparation (AutoPrep System): a. Centrifuge sample. b. Immunomagnetic Selection: Incubate with anti-EpCAM ferrofluid. c. Magnetic separation and transfer to magnest. d. Fixation and Permeabilization. e. Immunofluorescent Staining: Incubate with anti-CK-PE (epithelial marker), anti-CD45-APC (leukocyte marker), and DAPI (nuclear dye). f. Aspirate and resuspend in magnetic cartridge.
- CTC Enumeration (Analyzer System): a. Cartridge is scanned by a 4-color fluorescence microscope. b. CTC Definition: A nucleated cell (DAPI+) expressing CK (CK-PE+), lacking CD45 (CD45-APC-), with morphological features of a cell. c. The system presents candidate images for final review and classification by a trained technician.
Protocol 2: Serial CTC Monitoring for Treatment Response Assessment
Purpose: To assess changes in CTC count as an early indicator of therapeutic efficacy. Materials: As per Protocol 1. Requires scheduled blood draws. Procedure:
- Baseline Draw: Collect blood prior to Cycle 1 of new therapy.
- Follow-up Draws: Collect blood at standardized intervals (e.g., 3-4 weeks after Cycle 1, then every 2-3 cycles).
- Analysis: Process all samples identically using Protocol 1.
- Interpretation: a. Favorable Response: CTC count below the validated threshold (e.g., <5 for MBC/mCRPC, <3 for mCRC) at follow-up, or conversion from high (≥threshold) to low. b. Unfavorable Response: CTC count at or above threshold at follow-up, or conversion from low to high.
Visualization
Title: CTC Prognostic Validation Workflow
Title: CTC Count as a Predictive Biomarker
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for CellSearch CTC Enumeration Studies
| Item | Function in CTC Analysis |
|---|---|
| CellSave Preservative Tube | Blood collection tube containing EDTA and a cellular preservative. Stabilizes blood samples for up to 96h at RT, preventing clotting and preserving epitopes for accurate CTC enumeration. |
| CellSearch CTC Kit | Reagent kit for specific cancer type (e.g., Breast, Colorectal, Prostate). Contains anti-EpCAM coated ferrofluid for capture, and fluorescent stains (CK-PE/CD45-APC/DAPI) for CTC identification. |
| Anti-EpCAM Ferrofluid | Magnetic nanoparticles conjugated to EpCAM antibodies. Enriches epithelial cells from whole blood via immunomagnetic separation during the AutoPrep process. |
| CK-PE (Anti-Cytokeratin Phycoerythrin) | Fluorescent antibody against intracellular cytokeratins 8, 18, 19. Positive staining (PE signal) confirms the epithelial origin of a nucleated cell. |
| CD45-APC (Anti-Leukocyte Antigen) | Fluorescent antibody against the pan-leukocyte marker CD45. Negative staining (no APC signal) is required to exclude hematopoietic cells from CTC classification. |
| DAPI (4',6-diamidino-2-phenylindole) | DNA-binding fluorescent dye. Stains the nucleus of all nucleated cells. A positive DAPI signal is required to confirm an intact nucleus and for morphological analysis. |
| CellSearch AutoPrep System | Automated sample preparation system. Standardizes and controls the steps of immunomagnetic capture, staining, washing, and cartridge preparation to minimize inter-operator variability. |
| CellSearch Analyzer II | Semi-automated fluorescence microscope. Automatically scans the cartridge, captures images of fluorescent events, and presents galleries for final classification by a trained operator. |
This document provides detailed Application Notes and Protocols for the technical validation of the CellSearch system, a clinically validated, automated platform for the enumeration of Circulating Tumor Cells (CTCs) in whole blood from patients with metastatic carcinomas. Within the broader thesis on standardizing CTC enumeration for metastatic cancer research and drug development, establishing rigorous reproducibility, sensitivity, and specificity data is paramount. These metrics underpin the reliability of CTC counts as pharmacodynamic biomarkers, predictive indicators, and tools for monitoring disease progression.
The performance of the CellSearch system is defined by its analytical validity. The following tables summarize key quantitative validation data.
Table 1: Reproducibility and Precision Data
| Metric | Description | Typical Value | Condition |
|---|---|---|---|
| Inter-assay Precision (CV) | Variation between different runs, operators, days. | < 10% | For samples with ≥ 100 CTCs/7.5 mL |
| Intra-assay Precision (CV) | Variation within a single run. | < 15% | For samples with ~10 CTCs/7.5 mL |
| Total Error | Combined imprecision and bias. | ± 15% | Across validated cancer types (CRPC, MBC, mCRC) |
Table 2: Sensitivity and Specificity Data
| Metric | Description | Typical Value | Notes |
|---|---|---|---|
| Analytical Sensitivity (Limit of Detection - LoD) | Lowest concentration reliably distinguished from zero. | 1-2 CTCs / 7.5 mL blood | Established via spiking experiments with tumor cell lines. |
| Analytical Specificity | Ability to exclusively identify EpCAM+/CK+/CD45- cells. | > 99% | Based on leukocyte (CD45+) rejection. |
| Clinical Sensitivity | Proportion of metastatic cancer patients with ≥ 2 CTCs (baseline). | Varies by cancer type (e.g., ~65% in mCRPC, ~50% in MBC) | Disease prevalence dependent. |
| Clinical Specificity | Proportion of healthy donors with < 2 CTCs. | ≥ 99.5% | Rare false positives from interfering substances or atypical cells. |
Detailed Experimental Protocols
Protocol for Determining Inter-Assay Reproducibility
Objective: To quantify the precision of the CellSearch CTC enumeration across multiple independent assay runs. Materials: CellSearch AccuCount samples (or patient samples with known high CTC count), CellSearch system, CellSave Preservative Tubes. Procedure:
- Sample Preparation: Aliquot a homogeneous, well-mixed patient sample or a spiked control sample (e.g., containing ~100 SKBR-3 cells) into 10 separate 7.5 mL volumes in CellSave tubes.
- Independent Processing: Process each of the 10 samples as separate assays on different days, preferably by two different qualified operators.
- CellSearch Analysis: Follow the manufacturer's IFU: a. Immunomagnetic enrichment with anti-EpCAM ferrofluid. b. Staining with anti-CK-PE (cytokeratin), anti-CD45-APC (leukocyte marker), and DAPI (nuclear dye). c. Automated fluorescence microscopy and presentation of candidate CTCs (CK+/DAPI+/CD45-).
- Enumeration & Calculation: For each sample, record the CTC count per 7.5 mL. Calculate the mean, standard deviation (SD), and coefficient of variation (CV% = (SD/mean)*100) across the 10 results.
Protocol for Determining Analytical Sensitivity (LoD)
Objective: To establish the minimum number of tumor cells reliably detected by the assay. Materials: Tumor cell line (e.g., SKBR-3, MCF-7), CellSearch Profile kits, blood from healthy donors, hematocytometer. Procedure:
- Cell Line Preparation: Culture and harvest tumor cells. Perform viability staining (e.g., Trypan Blue) and count using a hematocytometer.
- Spiking: Spike known, low numbers of tumor cells (e.g., 0, 1, 2, 5, 10 cells) into 7.5 mL of healthy donor blood in CellSave tubes. Prepare a minimum of 10 replicates per spike level.
- Processing & Analysis: Process all spiked samples through the complete CellSearch CTC profile assay.
- Data Analysis: Determine the recovery rate (Number of cells detected / Number of cells spiked) for each level. The LoD is defined as the lowest spike concentration where ≥ 90% of replicates return a positive count (≥1 cell), with a CV of ≤ 30%.
Protocol for Assessing Specificity via Leukocyte Exclusion
Objective: To validate the specificity of CTC identification by quantifying false-positive events from leukocytes. Materials: Blood from healthy donors (n≥50), CellSearch CTC kit. Procedure:
- Sample Collection: Draw blood from healthy volunteers directly into CellSave tubes.
- Routine Processing: Process each sample through the standard CellSearch CTC assay.
- Image Review: System software presents galleries of candidate events staining positive for DAPI and/or CK. The analyst manually verifies each event, rejecting any event that is CD45+ (bright APC signal) or lacks clear morphological features of a tumor cell.
- Calculation: Specificity is calculated as: (Number of samples with < 2 CTCs / Total number of healthy donor samples) * 100%. Individual event specificity is calculated from the total number of CK+/DAPI+ events reviewed that are conclusively CD45-.
Visualizations
CellSearch CTC Enumeration Workflow
CTC Identification Decision Tree
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for CellSearch CTC Analysis
| Item | Function | Critical Notes |
|---|---|---|
| CellSave Preservation Tubes | Stabilizes blood sample for up to 96h, preventing degradation of EpCAM and minimizing leukocyte apoptosis. | Essential for pre-analytical standardization; required for clinical trials. |
| CellSearch CTC/CPC Kit | Disease-specific reagent kits (e.g., for Breast, Prostate, CRC). Contains anti-EpCAM ferrofluid, staining reagents (CK, CD45, DAPI), buffers, and sample tubes. | Kit must match cancer type; CPC kits include additional markers (e.g., CD34 for endothelial cells). |
| CellSearch Control Kit | Contains stabilized, human epithelial cells for system performance verification. Used in daily quality control. | Validates instrument optics, fluidics, and reagent performance. |
| CellSearch AccuCount Samples | Pre-spiked, lyophilized control samples with a defined range of tumor cells. Used for precision and recovery studies. | Critical for reproducibility testing and operator training. |
| CellSearch System | Automated sample processor (AutoPrep) and analyzer (MagNest) with integrated fluorescence microscope and image analysis software. | Provides standardized, walk-away automation from sample to result presentation. |
| Veridex CellSpotter Analyzer (or equivalent) | The computer workstation running the analysis software that presents candidate CTC galleries for final classification. | Manual review by a trained analyst is a mandatory step in the protocol. |
Application Notes
Circulating Tumor Cell (CTC) enumeration is a critical liquid biopsy tool in metastatic cancer research. The primary methodologies are immunomagnetic enrichment (CellSearch) and alternative physical property-based platforms (size, density, deformability). This analysis compares their technical specifications, clinical utility, and protocol intricacies within a thesis focused on the CellSearch system protocol.
Core Technology Comparison
CellSearch System (Veridex/Johnson & Johnson, Menarini Silicon Biosystems)
- Principle: Immunomagnetic positive selection targeting EpCAM (Epithelial Cell Adhesion Molecule).
- Enumeration: Immunofluorescent staining for cytokeratins (CK 8, 18, 19), CD45 (leukocyte exclusion), and DAPI (nuclear stain). A CTC is defined as CK+/DAPI+/CD45-.
- Regulatory Status: FDA-cleared for prognostic use in metastatic breast, colorectal, and prostate cancers.
- Output: Primarily an enumeration assay, with potential for subsequent molecular analysis from isolated cells (rarely performed in standard protocol).
Alternative Platforms (Size/Marker-Independent)
- Principles:
- Size-Based Filtration: Uses porous membranes (e.g., ISET, ScreenCell, Parsortix) to capture larger, rigid CTCs.
- Microfluidic-Based (Deformability): Uses label-free technologies like Dean Flow Fractionation (e.g., Vortex) or lateral displacement (e.g., ClearCell FX) based on cell size and deformability.
- Enumeration: Downstream immunofluorescence or morphological analysis is required for confirmation.
- Key Advantage: Captures EpCAM-low/negative CTCs (e.g., mesenchymal, from certain cancer types).
Table 1: Quantitative Platform Comparison
| Parameter | CellSearch | Size-Based Filtration (e.g., ISET) | Microfluidic Deformability (e.g., ClearCell FX) |
|---|---|---|---|
| Capture Principle | Positive selection (EpCAM) | Physical size/rigidity | Size & deformability |
| Typical Recovery Rate | 60-85% (EpCAM+ cells) | 50-80% (varies by size) | 60-90% (cell line dependent) |
| Purity of Enrichment | Low (<1% WBC depletion) | Very Low (high WBC retention) | Moderate (varies by system) |
| Sample Volume Processed | 7.5 mL (definitive) | 1-10 mL (flexible) | 2-8 mL (system dependent) |
| Throughput (samples/day) | ~24-30 | 4-10 (manual) | 12-16 |
| FDA Clearance | Yes (prognostic) | No (RUO/CE-IVD) | No (RUO) |
| Live Cell Retrieval | No (fixed) | Possible (depends on protocol) | Yes (key feature) |
| Key Limitation | EpCAM bias | Clogging, size bias | Pre-filtration often required |
Table 2: Clinical Performance Comparison in Metastatic Breast Cancer
| Platform | CTC Positivity Rate (≥5 CTCs/7.5mL) | CTC Heterogeneity Captured | Correlation with Prognosis |
|---|---|---|---|
| CellSearch | ~50-60% | Epithelial (E) to Mixed (E/M) | Strong, validated (≥5 CTCs = worse PFS/OS) |
| Size-Based | ~55-70% | Epithelial (E), Mixed (E/M), Mesenchymal (M) | Strong (thresholds vary) |
| Marker-Independent Microfluidic | ~60-75% | Epithelial (E), Mixed (E/M), Mesenchymal (M) | Emerging strong evidence |
Detailed Protocols
Protocol 1: Standard CellSearch CTC Enumeration Assay
This protocol forms the core of the broader thesis context.
I. Sample Preparation & Reagent Solutions
- CellSave Preservative Tubes: Contains EDTA and a cellular preservative. Maintains cell integrity for up to 96 hours. Function: Prevents coagulation and preserves epitopes.
- CellSearch CTC Kit: Includes ferrofluid nanoparticles conjugated to anti-EpCAM antibodies, staining reagents (anti-CK-PE, anti-CD45-APC), nuclear dye (DAPI), and permeabilization buffer.
- CellSearch Autoprep System: Automated sample processing unit.
- CellTracks Analyzer II: Semi-automated 4-color fluorescence microscope.
II. Step-by-Step Workflow
- Blood Draw: Collect 10 mL blood into CellSave tube. Invert gently 8 times. Store at room temperature (15-30°C).
- Sample Loading: Pipette 7.5 mL of blood into a centrifuge tube within the Autoprep system.
- Immunomagnetic Enrichment:
- System adds anti-EpCAM ferrofluid. Incubates with sample.
- Magnetic assembly separates labeled cells, followed by washing.
- Staining & Permeabilization:
- Enriched cells are transferred to a chamber (Magnest) and immobilized magnetically.
- Automated addition of permeabilization buffer, anti-CK-PE, anti-CD45-APC, and DAPI.
- Cartridge Assembly: The Magnest is sealed into a cartridge for analysis.
- Image Acquisition & Analysis:
- Cartridge is scanned by the CellTracks Analyzer II.
- Software presents galleries of potential CTCs (CK+/DAPI+/CD45- objects) for final technician review and enumeration.
III. Quality Control
- Use of control cell lines (e.g., SKBR-3) spiked into healthy donor blood.
- Daily system suitability tests are mandatory.
Protocol 2: CTC Capture Using Parsortix (Size-Based, Marker-Independent Platform)
I. Sample Preparation & Reagent Solutions
- Parsortix PR1 Cassette: Disposable microfluidic cassette with a stepped 6.5 µm gap for size-based capture. Function: Physically traps cells >~6.5 µm.
- Cell Separation Buffer (PBS + 0.1% BSA): Used for sample dilution and washing. Function: Maintains cell viability and reduces non-specific adhesion.
- Fixation Buffer (4% PFA) or Lysis Buffer: For downstream molecular analysis or immunofluorescence, respectively.
II. Step-by-Step Workflow
- Blood Processing: Centrifuge blood collection tube (e.g., EDTA). Dilute plasma/buffy coat fraction in Cell Separation Buffer.
- System Setup: Prime the Parsortix instrument with buffer. Install a new PR1 cassette.
- Sample Loading & Capture: Load diluted sample via syringe pump. Hydrodynamic force pushes cells through the cassette, capturing larger cells (CTCs, large WBCs).
- Washing: Flush cassette with buffer to remove unbound cells and debris.
- Harvest: Reverse fluid flow to release captured cells into a collection tube for live cell culture or apply positive pressure to transfer cells onto a microscope slide for staining.
- Downstream Analysis: Perform immunofluorescence (PanCK, CD45, DAPI) or nucleic acid extraction.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Platform | Function |
|---|---|---|
| CellSave Preservative Tubes | CellSearch | Maintains CTC integrity for delayed processing. |
| Anti-EpCAM Ferrofluid | CellSearch | Immunomagnetic enrichment of epithelial CTCs. |
| CK-PE / CD45-APC / DAPI Stain | CellSearch / General | Definitive identification of CTCs (CK+/CD45-/DAPI+). |
| Microfluidic Cassette (e.g., PR1) | Parsortix | Size-based capture of CTCs in a disposable format. |
| Cell Separation Buffer (PBS/BSA) | Parsortix, Vortex | Maintains cell viability during label-free processing. |
| Red Blood Cell Lysis Buffer | Many Filtration Platforms | Lyses RBCs to reduce sample volume and increase purity pre-processing. |
| Antibody Panels for EMT | All (Downstream) | Identify CTC heterogeneity (Epithelial: E-Cadherin; Mesenchymal: Vimentin, N-Cadherin). |
Visualizations
CellSearch CTC Enumeration Workflow
Alternative CTC Platform Workflow
CTC Capture Principle Classification
Application Notes: Expanding the Utility of CTCs in Metastatic Cancer
While the CellSearch system protocol remains the gold standard for CTC enumeration in metastatic breast, prostate, and colorectal cancers—providing validated prognostic value—the field is rapidly advancing beyond simple counting. The isolation of intact, viable CTCs presents a unique opportunity for real-time, minimally invasive longitudinal biomarker analysis. The following notes detail key applications.
Table 1: Molecular Analyses Derived from CTCs Beyond Enumeration
| Analysis Type | Key Target/Readout | Clinical/Research Utility |
|---|---|---|
| Protein Expression | HER2, AR, PD-L1, Ki67 | Assessment of therapeutic target status and tumor proliferative index. |
| Genomic Analysis | PIK3CA, ESR1, AR mutations; Copy Number Variations (CNVs) | Identification of actionable mutations and mechanisms of resistance. |
| Transcriptomic Profiling | mRNA sequencing (bulk/single CTC) | Classification of subtypes, epithelial-mesenchymal transition (EMT) status, and signaling pathway activity. |
| Functional Analysis | Ex vivo culture, drug sensitivity testing, mouse xenograft formation (CDX) | Direct evaluation of drug response and tumorigenic potential. |
Key Insight: Recent studies indicate that in hormone receptor-positive metastatic breast cancer, up to 40% of patients show discordance in ESR1 mutation status between CTC-derived DNA and archival primary tumor tissue, highlighting the critical need for real-time molecular profiling.
Detailed Protocols
Protocol 1: CTC Harvest for Downstream Molecular Analysis from CellSearch Cartridges
Principle: This protocol describes the retrieval of fixed CTCs from the CellSearch cartridge post-enumeration for fluorescence in situ hybridization (FISH) or immunofluorescence (IF) staining.
Materials (Research Reagent Solutions):
- CellSearch CXC Kit: For initial CTC enrichment and staining (EpCAM-coated ferrofluid, DAPI, anti-CK-PE, anti-CD45-APC).
- Permeabilization Buffer: (0.5% Triton X-100 in PBS) – Permeabilizes cell membranes for intracellular probe access.
- Hybridization Buffer: Specific to FISH probe set – Enables denaturation and hybridization of DNA probes.
- Formamide Wash Buffer: (2x SSC with formamide) – Provides stringent post-hybridization washing.
- Mounting Medium with DAPI: Preserves specimen and provides nuclear counterstain.
Procedure:
- Enumeration Completion: After completing the standard CellSearch enumeration protocol, carefully note the cartridge ID and the positions of CTCs.
- Cartridge Disassembly: Using a tool, gently pry open the analysis cartridge. The sample is contained on the interior surface of the cover.
- Spot Retrieval: The cover contains a defined "spot" area. Using a scalpel, carefully scrape the material from the spot into a 1.5 mL microcentrifuge tube containing 100 µL of PBS.
- Cytospin Preparation: Load the cell suspension into a cytocentrifuge funnel and centrifuge at 800 rpm for 5 minutes onto a glass slide.
- Slide Processing: Air-dry the slide for 1 hour. Proceed to FISH or multiplex IF protocols using the permeabilization and hybridization buffers as required.
Protocol 2: Isolation of Viable CTCs forEx VivoCulture Using an Adhesion-Free Platform
Principle: This method uses a negative selection and size-based filtration strategy to isolate unlabeled, viable CTCs from whole blood for culture attempts.
Materials (Research Reagent Solutions):
- RosetteSep CTC Depletion Cocktail: Antibody mixture for negative enrichment by depleting CD45+ leukocytes and CD235a+ erythrocytes.
- Ficoll-Paque Premium Density Gradient Medium: Separates mononuclear cells from granulocytes and erythrocytes.
- Cell Culture Medium: Specifically formulated (e.g., HITES for SCLC, organoid media for CRPC) with growth factors and Rho-associated kinase (ROCK) inhibitor.
- Ultra-Low Attachment Plate: Prevents adherence of normal stromal cells, selectively promoting growth of anchorage-independent CTC clusters.
Procedure:
- Blood Collection & Depletion: Draw blood into EDTA tubes. Mix with RosetteSep cocktail (50 µL/mL blood) and incubate for 20 minutes at room temperature.
- Density Gradient Centrifugation: Dilute the mixture 1:1 with PBS. Layer over Ficoll-Paque and centrifuge at 1200 × g for 20 minutes with the brake off.
- Mononuclear Cell Collection: Collect the mononuclear cell layer at the plasma-Ficoll interface.
- Wash & Plate: Wash cells twice in culture medium. Resuspend in complete culture medium supplemented with 10 µM Y-27632 (ROCK inhibitor) and seed into wells of an ultra-low attachment multi-well plate.
- Culture Monitoring: Maintain at 37°C, 5% CO₂. Monitor semi-weekly for sphere formation. Refresh half of the medium every 3-4 days.
Visualizations
Title: CTC Analysis Workflow from Enumeration to Characterization
Title: Key CTC-Derived Signaling Pathways & Drug Targets
The Scientist's Toolkit: Essential Reagents for CTC Characterization
Table 2: Key Research Reagent Solutions
| Reagent/Material | Function in CTC Analysis |
|---|---|
| CellSearch System Kits | FDA-cleared, standardized immunomagnetic enrichment and staining for CTC enumeration. |
| EpCAM-Coated Ferrofluid | Magnetic nanoparticle conjugate for positive selection of epithelial CTCs. |
| DAPI (4',6-diamidino-2-phenylindole) | Fluorescent nuclear stain for identifying nucleated cells. |
| Anti-Cytokeratin (CK) Antibodies (PE) | Fluorescent conjugates to identify epithelial origin of CTCs. |
| Anti-CD45 Antibodies (APC) | Fluorescent conjugates to identify and exclude leukocytes. |
| ROCK Inhibitor (Y-27632) | Critical supplement for ex vivo culture to inhibit anoids in single CTCs. |
| Whole Genome Amplification (WGA) Kit | Amplifies genomic DNA from single or few CTCs for downstream NGS. |
| SMART-seq Single-Cell RNA-seq Kits | For generating sequencing libraries from the low RNA yield of single CTCs. |
| Ultra-Low Attachment Cultureware | Supports growth of CTC clusters and organoids without stromal adherence. |
Integrating CellSearch Data into Clinical Trial Design and Drug Development Pathways
The enumeration and characterization of Circulating Tumor Cells (CTCs) via the CellSearch system have evolved from a prognostic biomarker to a cornerstone of modern drug development. This transition enables real-time, minimally invasive monitoring of tumor dynamics, offering a critical window into therapeutic efficacy, resistance mechanisms, and patient stratification. Integrating CellSearch-derived data into clinical trials is no longer adjunct but integral to accelerating and de-risking oncology drug development pathways.
Key Applications in Drug Development:
- Patient Stratification & Enrichment: Baseline CTC counts can identify high-risk metastatic populations, enriching trials for patients more likely to experience disease progression, thereby increasing the statistical power to detect a treatment effect.
- Pharmacodynamic (PD) Biomarker: Early on-treatment changes in CTC enumeration provide objective, rapid evidence of biological drug activity, often preceding radiographic changes.
- Surrogate Endpoint Exploration: Consistent correlation between CTC changes (e.g., conversion from unfavorable [≥5 CTCs/7.5mL] to favorable [<5 CTCs/7.5mL] at key timepoints) and Overall Survival (OS) supports its use as an intermediate endpoint for accelerated approval pathways.
- Understanding Resistance: Molecular characterization of captured CTCs (post-enumeration) can reveal emerging resistance pathways (e.g., AR-V7 in mCRPC), guiding subsequent therapy lines and combination strategies.
Quantitative Data Summary: Clinical Trial Impact of CTC Enumeration
Table 1: Correlation of CTC Count with Clinical Outcomes in Metastatic Cancers
| Cancer Type | Trial/Study Context | CTC Cut-off (per 7.5mL) | Key Finding (Outcome Correlation) | Reference (Example) |
|---|---|---|---|---|
| Metastatic Breast Cancer (MBC) | First-line therapy | Baseline ≥5 vs <5 | Shorter median PFS (4.9 vs 9.5 mos) and OS (17.5 vs >36 mos) for ≥5 | Cristofanilli et al., NEJM 2004 |
| Metastatic Castration-Resistant Prostate Cancer (mCRPC) | Post-chemotherapy (Abiraterone) | ≥5 at baseline | Independent predictor of lower radiographic PFS and OS | de Bono et al., Clin Cancer Res 2008; Scher et al., Lancet Oncol 2015 |
| Metastatic Colorectal Cancer (mCRC) | First-line therapy | ≥3 at baseline | Strong independent prognostic factor for reduced PFS and OS | Cohen et al., JCO 2008; Sastre et al., Ann Oncol 2012 |
| Non-Small Cell Lung Cancer (NSCLC) | First-line therapy | ≥5 at baseline | Associated with significantly worse PFS and OS | Krebs et al., J Thorac Oncol 2011 |
Table 2: CTCs as a Pharmacodynamic/Response Biomarker in Trials
| Trial Phase | Drug/Target | Cancer Type | CTC Measurement | Outcome Link |
|---|---|---|---|---|
| Phase III (SWOG S0500) | Chemotherapy Switch | MBC | CTCs at 1st follow-up (21 days) | Switching chemo for persistently high CTCs did not improve OS. Validated CTCs as a prognostic, not predictive, marker in this context. |
| Phase III (ELM-PC 4) | Abiraterone (CYP17 inhibitor) | mCRPC | CTC count at 13 weeks | CTC conversion to <5 coupled with PSA decline was a strong surrogate for OS benefit. |
| Phase II (Multiple) | Various Targeted Therapies | mCRPC, MBC | CTC enumeration & phenotypic characterization (e.g., AR-V7, HER2) | Changes in CTC subpopulations predict response to targeted agents (e.g., AR-V7+ linked to taxane over ARSi benefit). |
Detailed Experimental Protocols
Protocol 2.1: Standardized CellSearch CTC Enumeration for Clinical Trial Blood Samples
Purpose: To obtain reproducible, FDA-cleared enumeration of CTCs from whole blood samples in a multicenter trial setting.
Materials (The Scientist's Toolkit):
- CellSave Preservative Tubes: Contains EDTA and a cellular preservative. Function: Stabilizes blood cells and prevents epithelial cell degradation for up to 96 hours post-draw.
- CellSearch CTC Kit (Epithelial): Includes ferrofluid particles conjugated with anti-EpCAM antibodies, fluorescent nucleic acid dye (DAPI), anti-CK-PE (cytokeratin 8, 18, 19) antibodies, and anti-CD45-APC antibodies. Function: Immunomagnetically enriches epithelial cells and stains for identification (CK+, DAPI+, CD45-).
- CellSearch AutoPrep System: Automated sample preparation platform. Function: Standardizes all processing steps (immunomagnetic enrichment, staining, washing) to minimize inter-operator variability.
- CellSearch Magnest: Semi-automated fluorescence microscope cartridge loader. Function: Holds prepared sample cartridge for analysis.
- CellTrack Analyzer II: 4-color fluorescent microscope. Function: Digitally scans the entire cartridge surface, capturing images for each fluorescence channel.
- CellSearch CTC Classification Software: Applies predefined algorithms. Function: Identifies candidate events meeting size, morphology, and minimum fluorescence criteria (DAPI+, CK+) for final reviewer classification.
Workflow:
- Blood Draw & Shipment: Collect 10mL of whole blood directly into a CellSave tube. Invert gently 8 times. Store at room temperature (15-30°C). Ship to central lab within 96 hours.
- Sample Preparation (AutoPrep): a. Pipette 7.5 mL of blood into a centrifuge tube. b. Add ferrofluid anti-EpCAM particles. Incubate. EpCAM-expressing cells are bound to magnetic particles. c. Apply tube to magnetic separator. Discard supernatant. d. Resuspend cells. Transfer to a magnetically coated cartridge. e. Automatically stain with DAPI, anti-CK-PE, and anti-CD45-APC. f. Wash and permeabilize.
- Data Acquisition & Analysis: a. Place cartridge on the Magnest and load into the CellTrack Analyzer II. b. System scans ~300,000 image frames per cartridge. c. Software presents galleries of candidate events (objects meeting DAPI+ and CK+ thresholds) for reviewer confirmation. d. Reviewer classifies each candidate: A CTC is defined as DAPI+, CK-PE+ (within cell boundary), CD45-APC-, and exhibiting morphological features of a cell. All other events are rejected. e. Final CTC count per 7.5 mL blood is reported.
Protocol 2.2: Protocol for CTC-Based Patient Stratification in a Phase III Trial
Purpose: To stratify patients with metastatic breast cancer at baseline into "Favorable" vs. "Unfavorable" prognostic groups using CellSearch enumeration for trial enrollment or analysis stratification.
Methodology:
- Pre-Screening: Consent patients for biomarker sub-study.
- Baseline Blood Draw: Obtain blood in CellSave tube prior to Cycle 1, Day 1 of therapy.
- Centralized Processing: Ship samples to designated central laboratory for processing per Protocol 2.1.
- Stratification: Apply the predefined cut-off:
- Unfavorable Prognostic Group: ≥5 CTCs/7.5mL blood.
- Favorable Prognostic Group: <5 CTCs/7.5mL blood.
- Integration: Randomize patients within each CTC stratum or use the data as a stratification factor in the randomization algorithm. Alternatively, enroll only patients with ≥5 CTCs for a high-risk enrichment trial.
Protocol 2.3: Protocol for Longitudinal Pharmacodynamic Monitoring
Purpose: To assess early biological activity of a novel therapeutic by monitoring changes in CTC counts at predefined early timepoints.
Methodology:
- Timepoints: Schedule blood draws at:
- C1D1 (Baseline): Pre-dose.
- C1D15 or C2D1 (Early PD): 1-4 weeks after therapy initiation.
- Radiographic Assessment Timepoints: Align with standard-of-care imaging (e.g., every 8-12 weeks).
- Processing & Analysis: Process all samples identically per Protocol 2.1.
- Response Definition: Define a "CTC response":
- CTC Conversion: Change from ≥5 CTCs (unfavorable) at baseline to <5 CTCs (favorable) at the first follow-up.
- CTC Count Change: Percentage change from baseline (requires statistical pre-specification of meaningful threshold, e.g., ≥30% decline).
- Correlative Analysis: Correlate CTC response metrics (e.g., conversion rate at C2D1) with subsequent radiographic PFS and OS using Kaplan-Meier and Cox proportional hazards models.
Visualizations: Pathways and Workflows
Title: CTC Lifecycle and Therapeutic Impact Pathway
Title: CellSearch CTC Enumeration Workflow
Title: CTC-Guided Clinical Trial Design Logic
Conclusion
The CellSearch system protocol remains the most rigorously validated and clinically established method for CTC enumeration in metastatic cancer, providing a standardized, reproducible metric with proven prognostic and predictive utility. Its strengths lie in its regulatory status, robust automation, and extensive clinical evidence base, making it indispensable for translational research and clinical trials. However, the field is evolving, with new technologies aiming to capture EpCAM-negative CTCs and enable deeper molecular profiling. The future lies not in replacing CellSearch but in integrating its reliable enumeration data with complementary liquid biopsy approaches. For researchers and drug developers, mastering this protocol is fundamental, as CTC counts continue to serve as a critical pharmacodynamic biomarker and a window into metastatic biology, guiding personalized treatment strategies and accelerating therapeutic innovation.